Summary
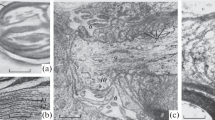
The hydrostatic (L p ) and osmotic (L PD ) filtration coefficients and the efflux rates of tritiated water were measured in the giant axon ofLoligo vulgaris. TheL p was 8 to 14×10−8 cm/sec/cm H2O and theL PD was two orders of magnitude smaller (3 to 6×10−10 cm/sec/cm H2O). In axons whose diameter was ∼500 μ, the time (t 1/2) required for a reduction in the axonal labeled water activity to one half its initial value was 38 to 48 sec. The rate limiting structure for solute flux was made ineffective by (1) storing the axon in isosmotic KF at 0–2 °C for one month to one year or by (2) fixing the axon in 2–4% glutaraldehyde for 3 to 7 hr. The criteria of ineffectiveness of the rate limiting structure for solute flux were (1) a reduction ofL PD to immeasurably low values, (2) the absence of electrical properties characteristic of a plasmalemma, and (3) a marked increase in the rate of efflux of Na22. In such impaired axons theL p and thet 1/2 of tritiated water efflux were unaffected. This independence of solute and solvent flux in conjunction with the finding that the hydraulic conductivity determined by bulk osmotic and hydrostatic pressure gradients is not equivalent (i.e.,L PD /L P ≪1) indicate that the rate limiting structures for solute and solvent flux are in series. Solvent fluxes appear to be surface-limited, not bulk-limited. We have been unable to resolve whether the surface structure involved in limiting solvent flux is the sheath (Schwann layer and adhering connective tissue) and/or the cortical layer of the axoplasmic gel.
Similar content being viewed by others
References
Baker, P.F., Hodgkin, A.L., Shaw, T.I. 1962. Replacement of the axoplasm of giant nerve fibres with artificial solutions.J. Physiol. (London) 164:330
Barnes, H. 1959. Apparatus and Methods of Oceanography. pp. 312–318, tables 33–39. Interscience Publishers, New York
Bear, R.S., Schmitt, F.O., Young, J.Z. 1937. Investigations on the protein constituents of nerve axoplasm.Proc. R. Soc. (London) B 123:520
Dick, D.A.T. 1964. The permeability coefficient of water in cell membrane and the diffusion coefficient in the cell interior.J. Theoret. Biol. 7:504
Hayes, R.M. 1968. A new proposal for the action of vasopressin based on studies of a complex synthetic membrane.J. Gen. Physiol. 51:385
Ling, G.N., Ochsenfeld, M.M., Karreman, G. 1967. Is the cell membrane a universal rate-limiting barrier to the movement of water between the living cell and its surrounding medium?J. Gen. Physiol. 50:1807
Løvthrup, S. 1963. On the rate of water exchange across the surface of animal cells.J. Theoret. Biol. 5:341
Nevis, A.H. 1958. Water transport in inverterbrate nerve fibres.J. Gen. Physiol. 41:927
Spyropoulos, C.S. 1960. Cytoplasmic pH of nerve fibres.J. Neurochem. 5:185
Spyropoulos, C.S. 1972. Some observations on the electrical properties of biological membranes.In: Membranes. A series of Advances. G. Eisenman, editor. p. 1267. Marcell Dekker, New York
Spyropoulos, C.S. 1977. Osmotic relations of nerve fiber.J. Membrane Biol. 32:19
Tasaki, I., Teorell, T., Spyropoulos, C.S. 1961. Movement of radioactive traces across squid axon membrane.Am. J. Physiol. 200:11
Vargas, F.F. 1968. Filtration coefficients of the axon membrane as measured by hydrostatic and osmotic methods.J. Gen. Physiol. 51:13
Villegas, R., Villegas, G.M. 1960. Membranes in nerve fiber of squid.J. Gen. Physiol. 43:73
White, M.L. 1960. The permeability of an acrylamide polymer gel.J. Phys. Chem. 64:1563
Author information
Authors and Affiliations
Additional information
The early experiments were undertaken in the Stazione Zoologica, Naples, and the Democritus Nuclear Research Center, Athens, Greece; the more recent experiments were undertaken in facilities provided by Prof. N. Nikolaou, Aegina, Greece.
Rights and permissions
About this article
Cite this article
Spyropoulos, C.S. Water fluxes in nerve fiber. J. Membrain Biol. 32, 1–18 (1977). https://doi.org/10.1007/BF01905206
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01905206