Abstract
The calcifying activities of biological fluids can be measured,in vitro andin vivo, by their ability to reharden softened blocks of tooth enamel. The activity is expressed in terms of the calcium (and phosphate) concentrations of synthetic calcifying solutions of known concentration and activity. A typical human plasma had an activity corresponding to that of a solution of the following concentration: calcium, 0.70 mM; Ca/P, 1.67 and fluoride, 0.05 mM.
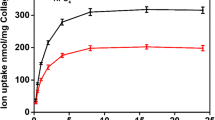
The relatively low activity of serums and plasmas was shown to arise from the presence of a number of ionic inhibitors. Pyrophosphate ion is one such inhibitor but cannot account for the major inhibition. Both anionic and cationic inhibitors were shown to be present, with the cations respresenting the major portion. The following ions at their reported physiological concentration were shown to be inhibitors in synthetic systems: P7O 4−7 , HCO −3 , SiO 2−4 , CrO 2−4 , Mg2+, Zn2+. In combination, these ions caused a total inhibition similar to those for plasmas.
Résumé
Les propriétés d'induction de calcification des liquides biologiques peuvent être mesurées, in vitro et in vivo, par leur possibilité de redurcir des blocs d'émail décalcifié. Cette propriété est exprimée par les concentrations de calcium (et de phosphate) de solutions synthétiques inductrices de calcification, de concentrations et d'activité connues. Un plasma humain typique a une activité inductrice correspondant à celle d'une solution contenant du calcium, 0,70 mM, Ca/P, 1,67 et fluorure, 0,05 mM.
L'activité relativement peu élevée des sérums et des plasmas s'explique par la présence d'inhibiteurs ioniques. L'ion phosphate est l'un d'eux, mais ne peut rendre compte de l'inhibition totale. Des anions et des cations sont responsables de l'inhibition, avec un rôle majeur pour les cations. Les ions suivants, en concentration physiologique, jouent un rôle inhibiteur: P7O 4−7 , HCO −3 , SiO 2−4 , CrO 2−4 , Mg2+, Zn2+. Un mélange de ces ions provoque une inhibition totale identique à celle du plasma.
Zusammenfassung
Der Einfluß biologischer Flüssigkeiten auf den Verkalkungsvorgang kann in vitro und in vivo anhand ihrer Fähigkeit, enthärtete Blöcke von Zahnschmelz wieder zu härten, gemessen werden. Diese Aktivität wird ausgedrückt als Konzentration des Calciums (und Phosphates) synthetischer calcifizierender Lösungen mit bekannter Konzentration und Aktivität. Die Aktivität eines charakteristischen menschlichen Plasmas entsprach derjenigen einer Lösung folgender Zusammensetzung: Calcium 0,70 mM; Ca/P=1,67 und Fluorid 0,05 mM.
Die relativ niedrige Aktivität von Serum und Plasma ist bedingt durch das Vorhandensein einer Anzahl ionischer Inhibitoren. Das Pyrophosphation ist ein solcher; er kann aber nicht für die gesamte Inhibition verantwortlich gemacht werden. Sowohl anionische als auch kationische Inhibitoren sind vorhanden, wobei die Kationen den Hauptanteil ausmachen. Folgende Ionen erwiesen sich in einer physiologischen Konzentration als Inhibitoren synthetischer Systeme: P7O 3−7 , HCO −3 , SiO 1−4 , CrO 2−4 , Mg2+, Zn2+. Zusammengenommen verursachten diese Ionen eine Gesamtinhibition ähnlich derjenigen des Plasmas.
Similar content being viewed by others
References
Bachra, B. N., Trautz, O. R., Simon, S. L.: The effect of magnesium and fluoride ions on the spontaneous precipitation of calcium carbonates and phosphates. Arch. oral Biol.10, 731–738 (1965).
Bird, E. D., Thomas, Jr., W.: Effect of various metals on mineralization in vitro. Proc. Soc. exp. Biol. (N.Y.)112, 640–643 (1963).
Brudevold F., Stedman, L. T., Smith, F. A.: Inorganic and organic components of tooth structure. Ann. N. Y. Acad. Sci.85, 110–132 (1960).
Campbell, D. O., Kilpatrick, M.: A kinetic study of the hydrolysis of pyrophosphates. J. Amer. Chem. Soc.76, 893–901 (1954).
Eisenstein, R., Ayers, J. P., Hass, G. M., Ellis, H.: Mineral binding by human arterial elastic tissue. Lab. Invest.13, 1198–1204 (1964).
Elliot, J. C.: Interpretation of carbonate bonds in infrared spectrum of dental enamel. J. dent. Res.42, 1081 (1963).
Ettori, J., Scoggan, S. M.: Ionized calcium in biological media. Nature (Lond.)184, 1315–1316 (1959).
Feagin, F.: A quantitative method for the measurement of factors affecting calcification processes. Ph. D. Thesis, University of Alabama, 1969.
Feagin, F.: Koulourides, T., Pigman, W.: The characterization of enamel surface demineralization, remineralization and associated hardness changes (Arch. oral. Biol. 1969, in press.)
Fleisch, H., Bisaz, S.: Isolation from urine of pyrophosphate, a calcification inhibitor. Amer. J. Physiol.203, 671–675 (1962).
— Neuman, W. F.: Mechanisms of calcification: Role of collagen polyphosphates and phosphatase. Amer. J. Physiol.200, 1296–1300 (1961).
— Russell, R. G. G., Bisaz, S., Termine, J. D., Posner, A. S.: Influence of pyrophosphate on the transformation of amorphous to crystalline calcium phosphate. Calc. Tiss. Res.2, 49–59 (1968).
— Straumann, F.: Effect of pyrophosphate on hydroxyapatite and its implication in calcium homeostasis. Nature (Lond.)212, 901–903 (1966a).
— Schibler, D., Maerki, J., Fossard, I.: Inhibition of aortic calcification by means of pyrophosphate and polyphosphates. Nature (Lond.)207, 1300–1301 (1965).
— Straumann, F., Schenk, R., Bisaz, S., Allgoewer, M.: Effect of condensed phosphates on calcification of chick embryo femurs in tissue culture. Amer. J. Physiol.211, 821–825 (1966b).
Glimcher, M. J.: Specificity of the molecular structure of organic matrices in mineralization. In: Calcification in biological systems, p. 421–487, ed. by Sognnaes, R. F.. Washington, D. C.: AAAS 1960.
Haumont, S.: Distribution of zinc in bone tissue. J. Histochem. Cytochem.9, 141–145 (1961).
Howard, J. E., Thomas, Jr., W. C., Barker, L. M., Smith, L. M., Wadkins, C. L.: The recognition and isolation from urine and serum of peptide inhibitors to calcification. Johns Hopk. med. J.120, 119–136 (1967).
Howland, J., Kramer, B.: Calcium and phosphorus in the serum in relation to rickets. Amer. J. Dis. Child.22, 105–119 (1921).
Koulourides, T., Feagin, F., Pigman, W.: Remineralization of dental enamel by saliva in vitro. Ann. N. Y. Acad. Sci.131, 771–775 (1965).
Little, M. F.: The effect of acid on enamel CO2. J. dent. Res.40, 903–914 (1961).
McLean, F. C., Hastings, A. B.: A biological method for the estimation of calcium ion concentration. J. biol. Chem.107, 337–350 (1934).
Mergenhagen, S. E., Martin, G. R., Rizzo, A. A., Wright, D. N., Scott, D. B.: Calcification in vivo of implanted collagen. Biochim. biophys. Acta (Amst.)43, 562–563 (1960).
Milhaud, G., Cherian, A. G., Moukhtar, M. S.: Calcium metabolism in the rat studied with calcium45: Effect of age. Proc. Soc. exp. Biol. (N. Y.)114, 382–384 (1963).
Neuman, W. F., Mulryan, B. J.: Synthetic hydroxyapatite crystals. III. The carbonate system. Calc. Tiss. Res.1, 94–104 (1967).
— Neuman, M. W.: The chemical dynamics of bone mineral. Chicago, Illinois: University of Chicago Press 1958.
Pigman, W., Kotwal, K., Koulourides, T.: Calcifying ability of human body fluids. Arch oral Biol.11, 815–824 (1966).
Rogers, H. J., Weidmann, S. M., Parkinson, A.: The collagen content of bones from rabbits, oxen, and humans. Biochem. J.50, 537–542 (1952).
Sadasivan, V.: Effect of feeding zinc on the liver and bones of rats. Biochem. J.48, 527–530 (1951).
Schibler, D., Fleisch, H.: Inhibition of skin calcification (calciphylaxis) by polyphosphates. Experientia (Basel)22, 367–369 (1966).
Sobel, A. E., Hanok, A.: Reversible inactivation of calcification in vitro and related studies. J. biol. Chem.197, 669–685 (1952).
Solomons, C. C., Neuman, W. F.: The remineralization of dentin. J. biol. Chem.235, 2502–2506 (1960).
Speirs, R. L., Spinelli, M., Brudevold, F.: Solution rate of hydroxyapatite in acetate buffer containing low concentrations of foreign ions. J. dent. Res.42, 811–820 (1963).
Trautz, O. R., Zapanta, R. R.: Experiments with calcium carbonate phosphates and the effect of topical application of sodium fluoride. Arch. oral Biol.4, 122–133 (1961).
Underwood, J. E.: Trace elements in human and animal nutrition. New York, N. Y.: Academic Press 1963.
Urist, M. R., Moss, M. J., Adams, J. M.: Calcification of tendon. Arch. Path.77, 594–608 (1964).
Wazer, J. van: Phosphorus and its compounds, vol. I. p. 452–459. New York N. Y.: Interscience Publ. Co., Inc. 1961.
Wadkins, C. L.: Experimental factors that influence collagen calcification in vitro. Calc. Tiss. Res.2, 214–228 (1968).
Author information
Authors and Affiliations
Additional information
This work has been supported by a grant from the American Chicle Company, Division of Warner-Lambert Pharmaceutical Company.
Rights and permissions
About this article
Cite this article
Feagin, F.F., Walker, A.A. & Pigman, W. Evaluation of the calcifying characteristics of biological fluids and inhibitors of calcification. Calc. Tis Res. 4, 231–244 (1969). https://doi.org/10.1007/BF02279126
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02279126