Abstract
The elongation growth of etiolated hypocotyl segments of lupin (Lupinus albus L.) was stimulated by acid pH (4.6 versus 6.5) and by IAA for periods of up to 4 h. After this time, the segments were unable to grow further. In the presence of an optimal IAA concentration (10 μM), acid pH increased the growth rate but had no effect on final growth. With suboptimal IAA (0.1 μM), however, acid pH increased growth in a more than additive way, suggesting a synergistic action between the two factors. This synergism may be explained by the increased IAA uptake and decarboxylation seen at an acid pH. These results reinforce the view that the effects of low pH and IAA on growth are not independent. Vanadate inhibited growth and also IAA uptake and decarboxylation. This inhibitor, therefore, probably inhibits growth not only by decreasing ATPase-mediated acidification but also by decreasing H+-dependent IAA uptake from the apoplasm. This dependence of IAA uptake on ATPase may be mediated by apoplasmic acidification. The amount of IAA decarboxylated increased when the assay conditions favored the growth of segments, indicating that IAA could be destroyed by decarboxylation during the auxin-induced growth.
Similar content being viewed by others
Abbreviations
- AIG:
-
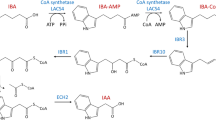
auxin (IAA)-induced growth
References
Behringer, F.J., Davies, P.J. (1992) Indole-3-acetic acid levels after phytochrome-mediated changes in the stem elongation rate of dark- and light-grown Pisum seedlings. Planta 188, 85–92
Bowman, B.J., Slayman, C.W. (1979) The effect of vanadate on the plasma membrane ATPase of Neurospora crassa. J. Biol. Chem. 254, 2128–2934
Brennan, T., Jacobs, P.J. (1983) In vivo measurement of indole-3-acetic acid decarboxylation in aging Coleus petiole sections. Plant Physiol. 73, 1–5
Brummell, D.A., Hall, J.L., Armstrong, C.F. (1986) Effect of vanadate on microsomal ATPase activity, acidification of the medium and auxin-stimulated growth in pea and cucumber. Ann. Bot. 57, 727–735
Cleland, R.E. (1971) Cell wall extension. Anu. Rev. Plant Physiol. 22, 197–222
Cleland, R.E. (1972) The dosage response curve for auxin-induced cell elongation: a reevaluation. Planta 104, 1–9
Cleland, R.E. (1987) Auxin and cell elongation. In: Plant hormones and their role in plant growth and development, pp. 132–148, Davies, P.J., ed. Kluwer Academic Publishers, Dordrecht Boston London
Cleland, R.E., Buckley, G., Nowbar, S., Lew, N.M., Stinemetz, C., Evans, M.L., Rayle, D.L. (1991) The pH profile for acidinduced elongation of coleoptile and epicotyl sections is consistent with the acid-growth theory. Planta 186, 70–74
Cohen, J.D., Bandurski, R.S. (1978) The bound auxins: protection of indole-3-acetic acid from peroxidase-catalyzed oxidation. Planta 70, 749–753
Davies, P.J., Rubery, P.H. (1978) Components of auxin transport in stem segments of Pisum sativum L. Planta 142, 211–219
Evans, M.L. (1974) Rapid responses to plant hormones. Annu. Rev. Plant Physiol. 25, 195–223
Galston, A.W., Dalberg, L. (1954) The adaptative formation and physiological significance of indoleacetic acid oxidase. Am. J. Bot. 41, 373–380
Goldsmith, M.H.M. (1977) The polar transport of auxin. Annu. Rev. Plant Physiol. 28, 439–478
Hager, A., Debus, G., Edel, H.-G., Stransky, H., Serrano, R. (1991) Auxin induces exocytosis and the rapid synthesis of a high-turnover pool of plasma-membrane H+-ATPase. Planta 157, 193–201
Hertel, R., Lomax, T.L., Briggs, W.R. (1983) Auxin transport in membrane vesicles Cucurbita pepo L. Planta 157, 193–201
Jacobs, M., Taiz, L. (1980) Vanadate inhibition of auxin-enhanced H+ secretion and elongation in pea epicotyls and oat coleoptiles. Proc. Natl. Acad. Sci. USA 77, 7242–7246
Jones, A.M., Cochran, D.S., Lamerson, P.M., Evans, M.L., Cohen, J.D. (1991) Red light-regulated growth I. Changes in the abundance of indoleacetic acid and a 22-kilodalton auxin-binding protein in the maize mesocotyl. Plant Physiol. 97, 352–358
Kutschera, U., Schopfer, P. (1985) Evidence against the acidgrowth theory of auxin action. Planta 163, 483–493
Lüthen, H., Bigdon, M., Böttger, M. (1990) Reexamination of the acid growth theory of auxin action. Plant Physiol. 93, 931–939
Ortuño, A., Sánchez-Bravo, J., Moral, J.R., Acosta, M., Sabater, F. (1990) Changes in the concentration of indole-3-acetic acid during the growth of etiolated lupin hypocotyls. Physiol. Plant. 78, 211–217
Penny, P., Penny, D. (1978) Rapid responses to phytohormones. In: Phytohormones and related compounds: a comprehensive treatise, vol. II, pp. 537–598, Letham, D.S., Goodwin, P.B., Higgins, T.J.V., eds. Elsevier/North-Holland Biomedical Press, Amsterdam Oxford New York
Rajagopal, R., Larsen, P. (1974) Auxin balance in the Avena coleoptile. Physiol. Plant. 31, 119–124
Rayle, D.L., Cleland, R.E. (1980) Evidence that auxin-induced growth of soybean hypocotyls involves proton excretion. Plant Physiol. 66, 433–437
Rayle, D.L., Cleland, R.E. (1992) The acid growth theory of auxin-induced cell elongation is alive and well. Plant Physiol. 99, 1271–1274
Reinecke, D.M., Bandurski, R.S. (1987) Auxin biosynthesis and metabolism. In: Plant hormones and their role in plant growth and development, pp. 24–42, Davies, P.J., ed. Kluwer Academic Publishers, Dordrecht Boston London
Rubery, P.H. (1978) Hydrogen ion dependence of carrier-mediated auxin uptake by suspension cultured crown gall cells. Planta 144, 173–178
Sánchez-Bravo, J., Ortuño, A., Acosta, M., Sabater, F. (1989) Evaluation of indole-3-acetic acid decarboxylating activity in hypocotyl sections of etiolated Lupinus albus seedlings. J. Plant Physiol. 134, 517–523
Sánchez-Bravo, J., Ortuño, A., Botía, J.M., del Rio, J.A., Caballero, M., Acosta, M., Sabater, F. (1990) Identification of the metabolites of indole-3-acetic acid in growing hypocotyls of Lupinus albus. Plant Growth Regul. 9, 315–327
Sánchez-Bravo, J., Ortuño, A., Botia, J.M., Acosta, M., Sabater, F. (1991) Lateral diffusion of polarly transported indoleacetic acid and its role in the growth of Lupinus albus L. hypocotyls. Planta 185, 391–396
Sánchez-Bravo, J., Ortuño, A., Botia, J.M., Acosta, M., Sabater, F. (1992) The decrease in auxin polar transport down the lupin hypocotyl could produce the indole-3-acetic acid distribution responsible for the elongation growth pattern. Plant Physiol. 100, 108–114
Schneider, E.A., Wightman, F. (1978) Auxins. In: Phytohormones and related compounds: a comprehensive treatise, vol. I, pp. 29–105, Letham, D.S., Goodwin, P.B., Higgins, T.J.V., eds. Elsevier/North-Holland Biomedical Press, Amsterdam Oxford New York
Schopfer, P. (1989) pH-dependence of extension growth in Avena coleoptiles and its implications for the mechanism of auxin action. Plant Physiol. 90, 202–207
Sembdner, G., Gross, D., Liebisch, H.W., Schneider, G. (1981) Biosynthesis and metabolism of plant hormones. In: Encyclopedia of plant physiology N.S., vol. 9: Hormonal regulation of development I, pp. 281–444, Pirson, A., Zimmermam, M.H., eds. Springer-Verlag, Berlin Heidelberg New York
Taiz, L. (1984) Plant cell expansion: regulation of cell wall mechanical properties. Annu. Rev. Plant Physiol. 35, 585–657
Thibaud, J.-B., Davidian, J.-C., Sentenac, H., Soler, A., Grignon, C. (1988) H+ Cotransports in corn roots as related to the surface pH shift induced by active H+ excretion. Plant Physiol. 88, 1469–1473
Tomaszewski, M., Thimann, K.V. (1966) Interactions of phenolic acids, metallic ions and chelating agents on auxin-induced growth. Plant Physiol. 41, 1443–1454
Valpuesta, V., Quesada, M.A., Sánchez-Roldán, C., Tigier, H., Heredia, A., Bukovac, M.J. (1989) Changes in indole-3-acetic acid, indole-3-acetic acid oxidase, and peroxidase isoenzymes in the seeds of developing peach fruits. J. Plant Growth Regul. 8, 255–261
Waldrum, J., Davies, E. (1981) Subcellular localization of IAA-oxidase in peas. Plant Physiol. 68, 1303–1307
Yamamoto, R., Maki, K., Yamagata, Y., Masuda, Y. (1974) Auxin and hydrogen ions actions on light-grown pea epicotyl segments I. Tissue specificity of auxin and hydrogen ion actions. Plant Cell Physiol. 15, 823–831
Author information
Authors and Affiliations
Additional information
We are grateful to Prof. R. Serrano (Dept. de Biotecnología, Universidad Politécnica, Valencia, Spain) for his critical comments and helpful suggestions, and to P. Thomas for proofreading the manuscript.
Rights and permissions
About this article
Cite this article
Botía, J.M., Ortuno, A., Sabater, F. et al. Uptake and decarboxylation of indole-3-acetic acid during auxin-induced growth in lupin hypocotyl segments. Planta 193, 224–231 (1994). https://doi.org/10.1007/BF00192534
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00192534