Summary
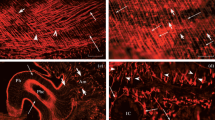
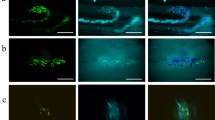
A concentration-dependent localization of octopamine-sensitive adenylate cyclase activity has been demonstrated in skeletal muscle of the locust, Schistocerca gregaria, using an histochemical technique. In the intermediate speed contracting muscle fibres from the fan region of the extensor-tibiae muscle of the locust hindleg, low concentrations of dl-octopamine (10−8 M) induce reaction product preferentially in the sarcoplasmic reticular component of the dyads. At slightly higher concentrations (10−7 and 10−6 M) lower amounts of diffuse reaction product are also found in the non-dyad sarcoplasmic reticulum and at the sarcolemmal membrane, with occassional amounts of a less diffuse, punctate product in the transverse tubule (T-tubule) component of the dyads. At higher concentrations (10−5 and 10−3 M) the predominant product is the dense, plaque-like accumulations of reaction product in the T-tubule component of the dyads. The results are discussed in terms of the likely physiological significance of the accumulation of reaction product in these different locations.
Similar content being viewed by others
References
Agnew WS (1987) Dual roles for DHP receptors in excitation-contraction coupling? Nature 328:297–298
Bowman WC (1981) Effects of adrenergic activation and inhibitors on the skeletal muscles. In: Handbook of Experimental Pharmacology. Vol 54: Szekeres L (ed) Adrenergic activators and inhibitors, part II. Springer, Berlin Heidelberg New York, pp 47–128
Bowman WC, Nott MW (1974) Effects of catecholamines, cyclic nucleotides and phosphodiesterase inhibitors on contraction of skeletal muscles in anaesthetized cats. Clin Exp Pharmacol Physiol 1:309–323
Caswell AH, Baker SP, Boyd H, Potter LT, Garcia M (1978) β-adrenergic receptor and adenylate cyclase in transverse tubules of skeletal muscle. J Biol Chem 253:3049–3054
Cutler LS (1983) Cytochemical methods for the localization of adenylate cyclase. A review and evaluation of the efficacy of the procedures. J Histochem Cytochem 31:85–93
Davenport AP, Evans PD (1984a) Stress-induced changes in the octopamine levels of insect haemolymph. Insect Biochem 14:135–143
Davenport AP, Evans PD (1984b) Changes in haemolymph octopamine levels associated with food deprivation in the locust, Schistocerca gregaria. Physiol Entomol 9:269–274
Downer RGH (1979a) Trehalose production in isolated fat body of the American cockroach, Periplaneta americana. Comp Biochem Physiol 62C:31–34
Downer RGH (1979b) Induction of hypertrehalosemia by excitation in Periplaneta americana. J Insect Physiol 25:59–63
Evans PD (1980) Biogenic amines in the insect nervous system. Adv Insect Physiol 15:317–473
Evans PD (1981) Multiple receptor types for octopamine in the locust. J Physiol (London) 318:99–122
Evans PD (1984a) A modulatory octopaminergic neurone increases cyclic nucleotide levels in locust skeletal muscle. J Physiol (London) 348:307–324
Evans PD (1984b) The role of cyclic nucleotides and calcium in the mediation of the modulatory effects of octopamine on locust skeletal muscle. J Physiol (London) 348:325–340
Evans PD (1984c) Studies on the mode of action of octopamine, 5-hydroxytryptamine and proctolin on a myogenic rhythm in the locust. J Exp Biol 110:231–251
Evans PD (1985a) Octopamine In: Kerkut GA, Gilbert L (eds) Comprehensive insect biochemistry, physiology and pharmacology. Pergamon Press, Oxford, pp 499–530
Evans PD (1985b) Regional differences in responsiveness to octopamine within a locust skeletal muscle. J Physiol (London) 366:331–341
Evans PD (1987) Phenyliminoimidazolidine derivatives activate both Octopamine 1 and Octopamine 2 receptor subtypes in locust skeletal muscle. J Exp Biol 129:239–250
Evans PD, Myers CM (1986) Peptidergic and aminergic modulation of insect skeletal muscle. J Exp Biol 124:143–176
Evans PD, O'Shea M (1977) An octopaminergic neurone modulates neuromuscular transmission in the locust. Nature 270:257–259
Gole JWD, Downer RGH (1979) Elevation of adenosine 3′,5′-monophosphate by octopamine in fat body of the American cockroach. Periplaneta americana L. Comp Biochem Physiol 64C:223–226
Howell SL, Whitfield M (1972) Cytochemical localization of adenylate cyclase activity in rat islets of Langerhans. J Histochem Cytochem 20:873–879
Hoyle G (1978) Distributions of nerve and muscle fibre types in locust jumping muscle. J Exp Biol 73:205–233
Jorgensen AO (1987) Immunolocalization of sarcoplasmic reticulum proteins in mammalian skeletal muscle fibers. Am Zool 27:1021–1032
Nakagawa M, Willner JH (1986) Adenylate cyclase in sarcoplasmic reticulum of skeletal muscle: Distribution, orientation and regulation. J Cyclic Nucleotide Protein Phosphor Res 11:237–251
Orchard I, Loughton BC, Webb RA (1981) Octopamine and short-term hyperlipaemia in the locust. Gen Comp Endocrinol 45:175–180
Raible DG, Cutler LS, Rodan GA (1978) Localization of adenylate cyclase in skeletal muscle sarcoplasmic reticulum and its relation to calcium accumulation. FEBS Lett 85:149–152
Rodan GA, Rodan SB, Raible DG, Cutler LS, Wacholtz M, Sha'afi RI (1979) Adenylate cyclase in muscular dystrophy. Ann NY Acad Sci 317:670–691
Schulze W, Krause E-G, Wollenberger A (1972) Cytochemical demonstration and localization of adenyl cyclase in skeletal and cardiac muscle. Adv Cyclic Nucleotide Protein Phosphor Res 1:249–260
Swales LS, Evans PD (1988) Histochemical localization of octopamine-and proctolin-sensitive adenylate cyclase activity in a locust skeletal muscle. Histochemistry 90:233–239
Vergara J, Asotra K (1987) The chemical transmission mechanism of excitation-contraction coupling in skeletal muscle. Physiol Sci 2:182–186
Vergara J, Tsien RY, Delay M (1985) Inositol 1,4,5-trisphosphate: a possible chemical link in excitation-contraction coupling in muscle. Proc Natl Acad Sci USA 82:6352–6356
Wagner RC, Kreiner P, Barnett RJ, Bitensky MW (1972) Biochemical characterization and cytochemical localization of a catecholamine-sensitive adenylate cyclase in isolated capillary endothelium. Proc Natl Acad Sci USA 69:3175–3179
Walaas SI, Horn RS, Nairn AC, Walaas O, Adler A (1988) Skeletal muscle sarcolemma proteins as targets for adenosine 3′:5′-monophosphate-dependent and calcium-dependent protein kinases. Arch Biochem Biophys 262:245–258
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Swales, L.S., Evans, P.D. A concentration-dependent localization of octopamine-sensitive adenylate cyclase activity in locust skeletal muscle. Histochemistry 92, 255–260 (1989). https://doi.org/10.1007/BF00500926
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00500926