Abstract
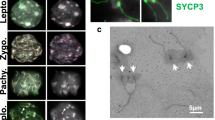
A transformed cell line, B1, of cerebral endothelial origin from the Wistar-Kyoto male rat has chromatid and chromosome type bridges in virtually every cell. It exhibits various dicentric and polycentric chromosomes. Most dicentrics are symmetric isochromosomes. Certain isodicentrics are present in a fair segment of the cell population; however, almost all cells have some newly arising isodicentrics. The live cells show a lengthened prometaphase. Anaphase is also retarded possibly due to the occurrence of bridges. At anaphase some multicentrics split at only one centromere. When pulled to the two poles the unsplit centromeres and the distal chromosome segment form a side arm bridge. Another mechanism appears to be a total lack of separation of daughter centromeres at meta-anaphase (‘meiotic-like’ behavior of mitotic chromosomes). This is realized by the pulling of each of the two unsplit centromeres to opposite poles and results in bridges with both sister chromatids running parallel to each other. A break at corresponding weak points in the two sister chromatids followed by rejoining can form a dicentric isochromosome. A third mechanism, the breakage-fusion-bridge cycle, is also operative but would not produce isodicentrics. In the case of the first two mechanisms some or all centromeres apparently split between telophase and onset of the following DNA synthesis rather than at the usual time at late metaphase. These observations may suggest some previously unknown behavior of multicentric chromosomes during mitosis.
Similar content being viewed by others
References
Diglio CA, Wolff DE, Meyers P (1983) Tranformation of rat cerebral endothelial cells by Rous Sarcoma virus. J Cell Biol 97:15–21
Dutrillaux B, Aurius A, Couturier S, Croquette W, Vegas-Pequignot E (1977) Multiple telomeric fusion and chain configurations in human somatic chromosomes. Chromosomes Today 6:37–45
Ernshaw WC, Migeon BR (1985) Three related centromere proteins are absent from the inactive centromere of a stable isodicentric chromosome. Chromosoma 92:290–296
Hepler PK (1980) Membranes in the mitotic apparatus of barley cells. J Cell Biol 86:490–499
Kovacs G, Muller-Brechlin R, Szucs S (1987) Telomeric association in two human renal tumors. Cancer Genet Cytogenet 28:363–366
McClintock B (1941) Spontaneous alterations in chromosome size and form in Zea mays. Cold Spring Harbor Symp Quant Biol 9:72–81
Merry DE, Pathak S, Hsu TC, Brinkley BR (1985) Antikinetochore antibodies: use as a probe for inactive centromeres. Am J Hum Genet 37:425–430
Parry P, Wolff S (1974) New Giemsa method for the differential staining of sister chromatids. Nature 251:156–158
Spurr AR (1969) A low viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26:31–43
Tyrkus M, Diglio CA, Gohle N (1983) Karyotype evolution in a transformed rat cerebral endothelial cell line. Int J Cancer 32:485–490
Vig BK (1984) Sequence of centromere separation: orderly segregation of multicentric chromosomes in mouse L-cells. Chromosoma 90:39–45
Vig BK, Zinkowski RP (1986) Sequence of centromere separation: a mechanism for orderly separation of dicentrics. Cancer Genet Cytogenet 23:347–359
Zinkowski RP, Vig BK, Broccoli D (1986) Characterization of kinetochores in multicentric chromosomes. Chromosoma 49:243–248
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vig, B.K., Paweletz, N. Sequence of centromere separation: generation of unstable multicentric chromosmes in a rat cell line. Chromosoma 96, 275–282 (1988). https://doi.org/10.1007/BF00286914
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00286914