Abstract
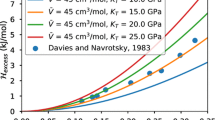
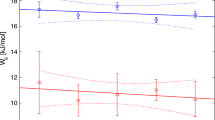
We have developed models for the thermody-namic properties of nephelines, kalsilites, and leucites in the simple system NaAlSiO4−KAlSiO4−Ca0.5AlSiO4−SiO2−H2O that are consistent with all known constraints on subsolidus equilibria and thermodynamic properties, and have integrated them into the existing MELTS software package. The model for nepheline is formulated for the simplifying assumptions that (1) a molecular mixing-type approximation describes changes in the configurational entropy associated with the coupled exchange substitutions □Si?NaAl and □Ca? Na2 and that (2) Na+ and K+ display long–range non-convergent ordering between a large cation and the three small cation sites in the Na4Al4Si4O16 formula unit. Notable features of the model include the prediction that the mineral tetrakalsilite (“panunzite”, sensu stricto) results from anti-ordering of Na and K between the large cation and the three small cation sites in the nepheline structure at high temperatures, an average dT/dP slope of about 55°/kbar for the reaction
over the temperature and pressure ranges 800–1050 °C and 500–5000 bars, roughly symmetric (i.e. quadratic) solution behavior of the K–Na substitution along joins between fully ordered components in nepheline, and large positive Gibbs energies for the nepheline reciprocal reactions
and
and for the leucite reciprocal reaction
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 24 April 1997 / Accepted: 21 August 1997
Rights and permissions
About this article
Cite this article
Sack, R., Ghiorso, M. Thermodynamics of feldspathoid solutions. Contrib Mineral Petrol 130, 256–274 (1998). https://doi.org/10.1007/s004100050364
Issue Date:
DOI: https://doi.org/10.1007/s004100050364