Summary
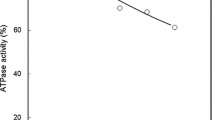
Myosins were isolated from dorsal ordinary muscles of carp acclimated to 10°C and 30°C for a minimum of 5 weeks and examined for their ATPase activities. Ca2+-ATPase activity was different between myosins from cold-and warm-acclimated carp, especially at KCl concentrations ranging from 0.1 to 0.2 M, when measured at pH 7.0. The highest activity was 0.32 μmol Pi·min-1·mg-1 at 0.2 M KCl for cold-acclimated carp and 0.47 μmol Pi·min-1·mg-1 at 0.1 M KCl for warm-acclimated fish. The pH-dependency of Ca2+-ATPase activity at 0.5 M KCl for both carp was, however, similar exhibiting two maxima around 0.3 μmol Pi·min-1·mg-1 at pH 6 and 0.4 μmol Pi·min-1·mg-1 at pH 9. K+(EDTA)-ATPase activity at pH 7.0 neither exhibited differences between both myosins. It increased with increasing KCl concentration showing the highest value of about 0.4 μmol Pi·min-1·mg-1 at 0.6–0.7 M KCl. Actin-activated myosin Mg2+-ATPase activity was markedly different between cold-and warm-acclimated carp. The maximum initial velocity was 0.53 μmol Pi·min-1·mg-1 myosin at pH 7.0 and 0.05 M KCl for cold-acclimated carp, which was 1.6 times as high as that for warm-acclimated carp. These differences were in good agreement with those obtained with myofibrillar Mg2+-ATPase activity between both carp. No differences were, however, observed in myosin affinity to actin. Differences in myosin properties between cold- and warm-acclimated carp were further evidenced by its thermal stability. The inactivation rate constant of myosin Ca2+-ATPase was 25·10-4·s-1 at 30°C and pH 7.0 for cold-acclimated carp, which was about 4 times as high as that for warm-acclimated carp. Light chain composition did not differ between both carp myosins. The differences in a primary structure of the heavy chain subunit was, however, clearly demonstrated between both myosins by peptide mapping.
Similar content being viewed by others
Abbreviations
- ATPase :
-
adenosine 5′-triphosphatase
- DTNB 5,5′:
-
dithio-bis-2-nitrobenzoic acid
- DTT :
-
dithiothreitol
- EGTA :
-
ethyleneglycol bis (β-aminoethylether)-N,N,N′,N′-tetraacetic acid
- K D :
-
inactivation rate constant
- SDS :
-
sodium dodecyl sulfate
- SDS-PAGE :
-
SDS-polyacrylamide gel electrophoresis
References
Cleveland DW, Fischer SG, Kirschner MW, Laemmli UK (1977) Peptide mapping by limited proteolysis in sodium dedecyl sulfate and analysis by gel electrophoresis. J Biol Chem 252:1102–1106
Ebashi S, Endo M (1968) Calcium and muscle contraction. Prog Biophys Mol Biol 18:123–183
Fiske CH, Subbarow Y (1925) The colorimetric determination of phosphorus. J Biol Chem 66:375–400
Goldspink G (1985) Malleability of the motor system. A comparative approach. J Exp Biol 115:375–391
Gornall AG, Bardawill CJ, David MM (1949) Determination of serum proteins by means of the biuret reaction. J Biol Chem 177:751–765
Guderley H, Blier P (1988) Thermal acclimation in fish conservative and labile properties of swimming muscle. Can J Zool 66:1105–1115
Hashimoto A, Kobayashi A, Arai K (1982) Thermostability of fish myofibrillar Ca-ATPase and adaptation to environmental temperatures. Bull Jpn Soc Sci Fish 48:671–684
Heap SP, Watt PW, Goldspink G (1985) Consequences of thermal change on the myofibrillar ATPase of five freswater teleosts. J Fish Biol 26:733–738
Heap SP, Watt PW, Goldspink G (1987) Contractile properties of goldfish fin muscles following temperature acclimation. J Comp Physiol B 157:219–225
Johnston IA (1979) Calcium regulatory proteins and temperature acclimation of actomyosin ATPase from a eurythermal teleost (Carassius auratus L.). J Comp Physiol B 129:163–167
Johnston IA, Walesby NJ (1977) Molecular mechanisms of temperature adaptation in fish myofibrillar adenosine triphosphatases. J Comp Physiol 119:195–206
Johnston IA, Davison W, Goldspink G (1975) Adaptations in Mg2+-activated myofibrillar ATPase activity induced by temperature acclimation. FEBS Lett 50:293–295
Johnston IA, Frearson N, Goldspink G (1973) The effects of environmental temperature on the properties of myofibrillar adenosine triphosphatase from various species of fish. Biochem J 133:735–738
Johnston IA, Walesby NJ, Davison W, Goldspink G (1977) Further studies on the adaptation of fish myofibrillar ATPases to different cell temperatures. Pflügers Arch 371:257–262
Kanoh S, Watabe S, Hashimoto K (1985) ATPase activity of requiem shark myosin. Bull Jpn Soc Sci Fish 51:973–977
Kohama K, Takano-Ohmuro H, Tanaka T, Yamaguchi Y, Kohama T (1986) Isolation and characterization of myosin from amoebae of Physarum polycephalum. J Biol Chem 261:8022–8027
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Ochiai Y, Kobayashi T, Watabe S, Hashimoto K (1990) Mapping of fish myosin light chains by two-dimensional gel electrophoresis. Comp Biochem Physiol 95B:341–345
Penney RK, Goldspink G (1981) Regulatory proteins and thermostability of myofibrillar ATPase in acclimated goldfish. Comp Biochem Physiol 69B:577–583
Somero GN (1975) Temperature as a selective factor in protein evolution. The adaptational strategy of “compromise”. J Exp Zool 194:175–188
Spudich JA, Watt S (1971) The regulation of rabbit skeletal muscle contraction. J Biol Chem 246:4866–4871
Stafford WF, Szentkiralyi EM, Szent-Györgyi AG (1979) Regulatory properties of single-headed fragments of scallop. Biochemistry 18:5273–5280
Tonomura Y (1972) Muscle proteins, muscle contraction and cation transport. University of Tokyo Press, Tokyo
Watabe S, Hashimoto K (1980) Myosins from white and dark muscles of mackerel. Some physico-chemical and enzymatic properties. J Biochem 87:1491–1499
Watabe S, Ochiai Y, Hashimoto K (1982) Identification of 5,5′-dithio-bis-2-nitrobenzoic acid (DTNB) and alkali light chains of piscine myosin. Bull Jpn Soc Sci Fish 48:827–832
Watabe S, Hashimoto K, Watanabe S (1983) The pH-dependency of myosin ATPases from yellowtail ordinary and dark muscles. J Biochem 94:1867–1875
Watabe S, Ushio H, Iwamoto M, Yamanaka H, Hashimoto K (1989) Temperature-dependency of rigor-mortis of fish muscle: Myofibrillar Mg2+-ATPase activity and Ca2+ uptake by sarcoplasmic reticulum. J Food Sci 54:1107–1110
Watabe S, Hwang GC, Ushio H, Hashimoto K (1990) Changes in rigor-mortis progress of carp induced by temperature acclimation. Agric Biol Chem 54:219–221
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hwang, G.C., Watabe, S. & Hashimoto, K. Changes in carp myosin ATPase induced by temperature acclimation. J Comp Physiol B 160, 233–239 (1990). https://doi.org/10.1007/BF00302588
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00302588