Summary
-
1.
Transepithelial potential differences (PDte) and acidification rates of the bath chamber were measured on isolated perfused posterior gills of the fiddler crabUca tangeri adapted to dilute seawater.
-
2.
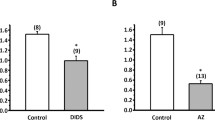
The PDte decreased to almost zero when Na+ was substituted by choline or when ouabain was added to the perfusion saline in high concentrations (10 mmol·l−1). Thus, the rheogenic NaCl-transport across the gill epithelium seems to be totally Na+-dependent.
-
3.
When Cl− was replaced by gluconate, a bath positive PDte occurred which was insensitive to ouabain. This PDte could also be observed when, in addition to Cl− removal, Na+ was replaced by TMA+.
-
4.
Bath acidification under normal conditions could be abolished by ouabain, indicating that there is H+ excretion via electrically silent Na+/H+ exchange. In contrast, bath acidification under Cl−-free conditions is only partially blocked by ouabain.
-
5.
It is concluded that under Cl−-free conditions a rheogenic H+-pump in the apical membrane is responsible for the ouabain-insensitive bath acidification as well as for the PDte.
Similar content being viewed by others
Abbreviations
- PD te :
-
transepithelial potential difference
- R te :
-
transepithelial resistance
- R in :
-
input resistance
- TMA + :
-
tetramethylammonium
- NEM :
-
N′-ethylmaleimide
References
Barra JA, Péqueux A, Humbert W (1983) A morphological study on gills of a crab acclimated to freshwater. Tissue and Cell 15:583–569
Bigalke T (1986) Charakterisierung der (Na+,K+)-ATPase und des Na+/H+-Austauschers aus dem ionentransportierenden Kiemenepithel der WollhandkrabbeEriocheir sinensis. Dissertation, FU Berlin
Burckhardt B-C, Sato K, Frömter E (1984) Electrophysiological analysis of bicarbonate permeation across the peritubular cell membrane of rat kidney proximal tubule. I. Basic observations. Pflügers Arch 401:34–42
Croghan PC, Curra RA, Lockwood APM (1965) The electrical potential difference across the epithelium of isolated gills of the crayfishAustropotamobius pallipes (Lerboullet). J Exp Biol 42:463–474
Drews G (1985) Elektrophysiologische und biochemische Untersuchungen zur osmoregulatorischen Fähigkeit und zur Salzaufnahme über das Kiemenepithel vonUca tangeri (Eydoux 1835). Dissertation, FU Berlin
Drews G, Graszynski K (1987) The transepithelial potential difference in the gills of the fiddler crab,Uca tangeri: influence of some inhibitors. J Comp Physiol B 157:345–353
Geck P, Pietrzyk C, Burckhardt B-C, Pfeiffer B, Heinz E (1980) Electrically silent cotransport of Na+,K+ and Cl− in Ehrlich cells. Biochim Biophys Acta 600:432–447
Gilles R, Péquex A (1986) Physiological and ultrastructural studies of NaCl transport in crustaceans gills. Boll Zool 53:173–182
Gocha N, Péqueux A, Wanson S, Gilles R (1987) Cl− fluxes across isolated, perfused gills of the chinese crabEriocheir sinensis (Milne Edwards) acclimated to freshwater. Comp Biochem Physiol 88A:581–584
Greger R (1981) Chloride reabsorption in the rabbit cortical thick ascending limb of the loop of Henle. A sodium dependent process. Pflügers Arch 390:38–43
Greger R, Schlatter E (1983a) Properties of the lumen membrane of the cortical thick ascending limb of Henle's loop of rabbit kidney. Pflügers Arch 396:315–324
Greger R, Schlatter E (1983b) Properties of the basolateral membrane of the cortical thick ascending limb of Henle's loop of rabbit kidney. A model for secondary active chloride transport. Pflügers Arch 396:325–334
Greger R, Schlatter E, Lang F (1983) Evidence for electroneutral sodium chloride cotransport in the cortical thick ascending limb of Henle's loop of rabbit kidney. Pflügers Arch 396:308–314
King EN, Schoffeniels E (1969) ‘In vitro’ preparation of crab gills for use in ion transport studies. Arch Int Physiol Biochim 77:105–111
Kinne-Saffran E, Beauwens R, Kinne R (1982) An ATP-driven proton pump in brush-border membranes from rat renal cortex. J Membr Biol 64:67–76
Kinsella JL, Aronson PS (1981) Amiloride inhibition of the Na+/H+ exchanger in renal microvillus membrane vesicles. Am J Physiol 241:F374-F379
Kirschner LB (1979) Control mechanisms in crustaceans and fishes. In: Gilles R (ed) Mechanisms of osmoregulation in animals. Wiley, Chichester New York
Krippeit-Drews P (1986) Elektrophysiologische Untersuchungen zum Ionentansport über das Kiemenepithel von hyperregulierenden Krabben der Gezeitenzone. Dissertation, FU Berlin
Krogh A (1938) The active absorption of ions in some freshwater animals. Zeitschr Vergl Physiol 25:335–350
Lucu C, Siebers D (1986) Amiloride-sensitive sodium flux and potentials in perfusedCarcinus gill preparations. J Exp Biol 122:25–35
Lucu C, Siebers D (1987) Linkage of Cl− fluxes with ouabain sensitive Na/K exchange throughCarcinus gill epithelia. Comp Biochem Physiol 87A:807–811
Murer H, Hopfer U, Kinne R (1976) Sodium/proton antiport in brush-border-membrane vesicles isolated from rat small intestine and kidney. Biochem J 154:579–604
Péquex A, Gilles R (1981) Na+-fluxes across isolated perfused gills of the Chinese crabEriocheir sinensis. J Exp Biol 92:173–186
Péqueux A, Gilles R (1988) The transepithelial potential difference of isolated perfused gills of the Chinese crabEriocheir sinensis acclimated to freshwater. Comp Biochem Physiol 89A:169–172
Sabolic I, Burckhardt G (1986) Characteristics of the proton pump in rat renal cortical endocytotic vesicles. Am J Physiol 250:F817-F826
Sabolic I, Haase W, Burckhardt G (1985) ATP-dependent H+-pump in membrane vesicles from rat kidney cortex. Am J Physiol 248:F835-F845
Schwartz GJ, Al-Awqati Q (1986) Regulation of transepithelial H+ transport by exocytosis. Annu Rev Physiol 48:153–161
Siebers D, Winkler A, Lucu C, Thedens G, Weichart D (1985) Na-K-ATPase generates an active transport potential in the gills of hyperregulating shore crabCarcinus maenas. Mar Biol 87:185–193
Siebers D, Lucu C, Winkler A, Dalla Venezia L, Wille H (1986) Active uptake of sodium in the gills of the hyperregulating shore crabCarcinus maenas. Helgol Wiss Meeresunters 40:151–160
Author information
Authors and Affiliations
Additional information
Parts of this study had been published earlier in abstract form
Rights and permissions
About this article
Cite this article
Krippeit-Drews, P., Drews, G. & Graszynski, K. Effects of ion substitution on the transepithelial potential difference of the gills of the fiddler crabUca tangeri: evidence for a H+-pump in the apical membrane. J Comp Physiol B 159, 43–49 (1989). https://doi.org/10.1007/BF00692682
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00692682