•
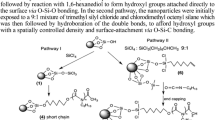
)4, though it is expected to relax via formation of peroxidic bridges), and the oxygen-bridge vacancy (which in the absence of relaxation can be described as a diradical center (≡Si•)2– the E′′ center). These radicals react with the residual atmosphere according to completely new pathways: the bombardment in a CO2 atmosphere results in the formation of ester-like and carboxylate groups, stable up to 500 °C at least, inserted in the SiO2 network at the oxygen-bridge vacancies. The bombardment in a C2H4 atmosphere results in more complex configurations: the oxygen-bridge vacancy reacts at room temperature with ethylene forming a Lewis adduct which, after heating at 500 °C, presumably reverts to a -CH2-CH2- bridge in between silicon atoms; the silicon-link vacancy likely reacts with C2H4 forming CH3CHO. These conclusions, based on experimental data (mainly infrared spectroscopy), are also supported by extended quantum mechanical calculations (density-functional methods and ab initio molecular dynamics).
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 6 March 1998/Accepted: 21 September 1998
Rights and permissions
About this article
Cite this article
Cerofolini, G., Meda, L. CO2 and C2H4 grafting to the native defects of ion-bombarded porous silica . Appl Phys A 68, 29–39 (1999). https://doi.org/10.1007/s003390050850
Issue Date:
DOI: https://doi.org/10.1007/s003390050850