Abstract
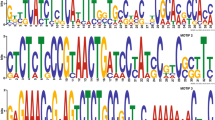
Based on conserved amino-acid regions predicted for the chitin synthases (Chs) of Saccharomyces cerevisiae, two different primer sets were synthesized and used in polymerase chain reactions (PCRs) to amplify 614-bp and 366-bp sequences from genomic DNA of the zoopathogenic fungus Phialophora verrucosa. DNA-sequencing and Southern-blotting analyses of the 614-bp DNA amplification products suggested that portions of two distinct P. verrucosa chitin synthase genes (PvCHS1, PvCHS2), coding for two different zymogenic-type PvChs isozymes, had been identified. The deduced amino-acid sequence of each fell into different Chs classes, namely class I and class II. In addition, the 366-bp DNA segment was shown to code for a conserved region having homology with the CSD2/CAL1 gene of S. cerevisiae, which encodes a nonzymogenic-type enzyme, Chs3, in that fungus. The amino-acid sequence derived from PvCHS3 exhibits 88.2% similarity and 78.4% identity to the same amino-acid region of the S. cerevisiae enzyme. These results provide a critical first step toward investigating the molecular and pathogenic importance of CHS gene regulation in this fungus and for exploring steps leading to Chs function as potential targets for the design of new therapeutic agents.
Similar content being viewed by others
References
Au-young J, Robbins PW (1990) Isolation of a chitin synthase (CHS1) from Candida albicans by expression in Saccharomyces cerevisiae. Mol Microbiol 4:197–207
Bowen AR, Chen-Wu JL, Momany M, Young R, Szaniszlo PJ, Robbins PW (1992) Classification of fungal chitin synthases. Proc Natl Acad Sci USA 89:519–523
Bulawa CE (1992) CSD2, CSD3, and CSD4, genes required for chitin synthesis in Saccharomyces cerevisiae: the CSD2 gene product is related to chitin synthases and to developmentally regulated proteins in Rhizobium species and Xenopus laevis. Mol Cell Biol 12:1764–1776
Bulawa CE, Slater M, Cabib E, Au-Young J, Sburlati A, Adair Jr. WL, Robbins PW (1986) The S. cerevisiae structural gene for chitin synthase is not required for chitin synthesis in vivo. Cell 46:213–225
Cabib E (1991) Differential inhibition of chitin synthetases 1 and 2 from Saccharomyces cerevisiae by polyoxin D and nikkomycins. Antimicrob Agents Chemother 35:170–173
Cabib E, Mol PC, Shaw JA, Choi WJ (1993) Biosynthesis of cell wall and septum during yeast growth. Arch Med Res 24:301–303
Chen-Wu J, Zwicker J, Bowen AR, Robbins PW (1992) Expression of chitin synthase genes during yeast and hyphal growth phases of Candida albicans. Mol Microbiol 6:497–502
Chua SS, Momany M, Mendoza L, Szaniszlo PJ (1994) Identification of three chitin synthase genes in the dimorphic fungal pathogen Sporothrix schenckii. Curr Microbiol 29:151–156
Churchill ACL, Ciufetti LA, Hansen DR, Van Etten HD, Van Alfen NK (1990) Transformation of the fungal pathogen Cryphonectria parasitica with a variety of heterologous plasmids. Curr Genet 17:25–31
Cooper BH (1985) Phialophora verrucosa and other chromoblastomycotic fungi. In: Szaniszlo PJ (ed) Fungal dimorphism with emphasis on fungi pathogenic for humans. Plenum Press, New York, pp 263–280
Cooper Jr. CR, Harris JL, Jacobs CW, Szaniszlo PJ (1984) Effect of polyoxin AL on cellular development in Wangiella dermatitidis. Exp Mycol 8:349–363
Fader RC, McGinnis MR (1988) Infections caused by dematiaceous fungi: chromoblastomycosis and phaehyphomycosis. Infect Dis Clin North Am 2:925–938
Hector RF (1993) Compounds active against cell walls of medically important fungi. Clin Microb Rev 6:1–21
Kwon-Chung KJ, Bennett JE (1992) Chromoblastomycosis. In: Kwon-Chung KJ, Bennett JE (eds) Medical mycology. Lea and Febiger, Philadelphia, pp 337–355
Mendoza L, Karuppayil SM, Szaniszlo PJ (1993) Calcium regulates in vitro dimorphism in chromoblastomycotic fungi. Mycoses 36:157–164
Miyazaki A, Momany M, Szaniszlo PJ, Jayaram M, Ootaki T (1993) Chitin synthase-encoding gene(s) of the zygomycete fungus Phycomyces blakesleeanus. Gene 134:129–134
McCullough JE (1992) Importance of chitin synthesis for fungal growth and as a target for antifungal agents. In: Fernandes PB (ed) New approaches for antifungal drugs. Birkhäuser, Boston, pp 32–45
Pan S, Sigler L, Cole GT (1994) Evidence for a phylogenetic connection between Coccidioides immitis and Uncinocarpus reesii (Onygenaceae). Microbiology, 140:1481–1494
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74:5463–5467
Shaw JA, Mol PC, Bowers B, Silverman SJ, Valdivieso MH, Duran A, Cabib E (1991) The function of chitin synthases 2 and 3 in the Saccharomyces cerevisiae cell cycle. J Cell Biol 114:111–123
Silverman SJ (1989) Similar and different domains of chitin synthase 1 and 2 of S. cerevisiae: two isozymes with distinct functions. Yeast 5:495–467
Silverman SJ, Sburlati A, Slater ML, Cabib E (1988) Chitin synthase 2 is essential for septum formation and cell division in Saccharomyces cerevisiae. Proc Natl Acad Sci USA 85:4735–4739
Sudoh M, Nagahashi S, Doi M, Ohta A, Takagi M, Arisawa M (1993) Cloning of the chitin synthase 3 gene from Canida albicans and its expression during yeast-hyphal transition. Mol Gen Genet 241:351–358
Szaniszlo PJ, Momany M (1993) Chitin, chitin synthase and chitin synthase-conserved-region homologues in Wangiella dermatitidis. In: Maresca B, Kobayashi GS, Yamaguchi H (eds) Molecular biology and its application to medical mycology. Springer Berlin Heidelberg, pp 229–242
Valdivieso MH, Mol PC, Shaw JA, Cabib E, Duran A (1991) CAL1, a gene required for activity of chitin synthase 3 in Saccharomyces cerevisiae. J Cell Biol 114:101–109
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Peng, M., Karuppayil, S.M., Mendoza, L. et al. Use of the polymerase chain reaction to identify coding sequences for chitin synthase isozymes in Phialophora verrucosa . Curr Genet 27, 517–523 (1995). https://doi.org/10.1007/BF00314441
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00314441