Summary
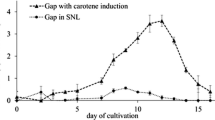
Extracellular culture filtrates from ligninolytic cultures of the lignin-degrading basidiomycete Lentinula (syn. Lentinus) edodes (Berk.) Pegler contained one major peroxidase when grown on a commercial oak-wood substrate. The peroxidase was purified by polyethylenimine clarification, anion-exchange chromatography, and hydrophobic-interaction HPLC. The enzyme (MnP1) was a heme-iron protein with an apparent molecular weight of 44 600 on sodium dodecyl sulfate-polyacrylamide gel electrophoresis gels and an isoelectric point of pH 3.2. The native enzyme had an absorption maximum at 407 nm, which shifted to 420 nm upon H2O2 addition. The pyridine-hemochrome-absorption spectrum indicated that one heme group was present per enzyme as protoporphyrin IX. N-terminal amino acid sequencing showed that MnP1 had higher sequence homology with manganese peroxidases than with lignin peroxidases reported from Phanerochaete chrysosporium. L. edodes MnP1 was capable of oxidizing lignin and lignin-model compounds in the presence of manganese and H2O2.
Similar content being viewed by others
References
Boer HA de, Zhang YZ, Collins C, Reddy CA (1987) Analysis and nucleotide sequence of two ligninase cDNAs from a white rot fungus, Phanerochaete chrysosporium. Gene 58:93–102
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chang ST, Miles PG (1989) Lentinus — a mushrooming mushroom. In: Edible mushrooms and their cultivation, CRC Press, Boca Raton pp 189–223
Forrester IT, Grabski AC, Burgess RR, Leatham GF (1988) Manganese, Mn-dependent peroxidases, and the biodegradation of lignin. Biochem Biophys Res Commun 157:992–999
Forrester IT, Grabski AC, Mishra C, Kelley BD, Strickland WN, Leatham GF, Burgess RR (1990) Purification and characterization of a manganese-dependent peroxidase from Lentinula edodes. In: Kirk TK, Chang H-M (eds) Applications of biotechnology in pulp and paper manufacture. Butterworths, Stoneham, Ma, USA (in press)
Glenn JK, Gold MH (1985) Purification and characterization of an extracellular Mn(II)-dependent peroxidase from the lignin-degrading basidiomycete, Phanerochaete chrysosporium. Arch Biochem Biophys 242:329–341
Glenn JK, Morgan MA, Mayfield MB, Kuwahara M, Gold MH (1983) An extracellular H2O2-requiring enzyme preparation involved in lignin biodegradation by the white rot basidiomycete Phanerochaete chrysosporium. Biochem Biophys Res Commun 114:1077–1083
Glenn JK, Akileswaran L, Gold MH (1986) Mn (II) oxidation is the principal function of the extracellular Mn peroxidase from Phanerochaete chrysosporium. Arch Biochem Biophys 251:688–696
Gold MH, Kuwahara M, Chiu AA, Glenn JK (1984) Purification and characterization of an extracellular diarylpropane oxygenase from the white-rot basidiomycete Phanerochaete chrysosporium. Arch Biochem Biophys 234:353–362
Gold MH, Wariishi H, Valli K (1989) Extracellular peroxidases involved in lignin degradation by Phanerochaete chrysosporium. In: Whitaker JR, Sonnet PF (eds) Biocatalysis in agricultural biotechnology, ACS Symposium Series 389. American Chemical Society, Washington, pp 127–140
Huynh V-B, Paszczyñski AJ, Olson P, Crawford R (1986) Oxidation of lignin model compounds by manganese-dependent enzymes from Phanerochaete chrysosporium as compared to chemically-generated Mn(III). In: Eriksson KE (ed) Proceedings of the Third International Conference on Biotechnology in the Pulp and Paper Industry, Stockholm, June 16–19, STFI/SPCI, Stockholm, pp 42–45
Jeffries TW, Choi S, Kirk TK (1981) Nutritional regulation of lignin degradation by Phanerochaete chrysosporium. Appl Environ Microbiol 42:290–296
Johansson T, Nyman PO (1987) A manganese(II)-dependent peroxidase from the white-rot fungus Trametes versicolor. Acta Chem Scand 41:762–765
Kuwahara M, Glenn JK, Morgan MA, Gold MH (1984) Separation and characterization of two extracellular H2O2-dependent oxidases from ligninolytic cultures of Phanerochaete chrysosporium. FEBS Lett 169:247–250
Leatham GF (1982) Cultivation of shiitake, the Japanese forest mushroom, on logs: a potential industry for the United States. For Prod J 32:29–35
Leatham GF (1985) Extracellular enzymes produced by the cultivated mushroom Lentinula edodes during degradation of a lignocellulosic medium. Appl Environ Microbiol 50:859–867
Leatham GF (1986) The ligninolytic activities of Lentinula edodes and Phanerochaete chrysosporium. Appl Microbiol Biotechnol 24:51–58
Leatham GF, Leonard TJ (1989) Biology and physiology of shiitake cultivation. In: Shiitake mushrooms: a national symposium and trade show. Alternative Crop Center, University of Minnesota, St. Paul, pp 19–28
Leatham GF, Stahmann MA (1981) Studies on the laccase of Lentinula edodes. Specificity, localization and association with the development of fruiting bodies. J Gen Microbiol 125:147–157
Leisola MSA, Kozulic B, Meusdoerffer F, Fiechter A (1987) Homology among multiple extracellular peroxidases from P. chrysosporium. J Biol Chem 262:419–424
Pajot P, Gooudinsky O (1970) Molecular weight and quarternary structure of yeast l-lactate dehydrogenase (cytochrome b 2). Revised heme extinction coefficients and minimal molecular weight. Eur J Biochem 12:158–164
Paszczyñski A, Huynh V-B, Crawford RL (1985) Enzymaticactivities of an extracellular, manganese-dependent peroxidase from Phanerochaete chrysosporium. FEMS Microbiol Lett 29:37–41
Paszczyñski A, Huynh V-B, Crawford RL (1986) Comparison of liginase-1 and peroxidase M-2 from the white-rot fungus Phanerochaete chrysosporium. Arch Biochem Biophys 244:750–765
Pease EA, Andrawis A, Tien M (1989) Manganese-dependent peroxidase from Phanerochaete chrysosporium primary structure deduced from cDNA sequence. J Biol Chem 264:13 531–13 535
Pribnow D, Mayfield MB, Nipper VJ, Brown JA, Gold MH (1989) Characterization of a cDNA encoding a manganese peroxidase, from the lignin-degrading basidiomycete Phanerochaete chrysosporium. J Biol Chem 264:5036–5040
Royse DJ, Schisler LC (1980) Mushrooms — their consumption, production, and culture development. Interdiscip Sci Rev 5:324–332
Schalch H, Gaskell J, Smith TL, Cullen D (1989) Molecular cloning and sequences of lignin peroxidase genes of Phanerochaete chrysosporium. Mol Cell Biol 9:2743–2747
Smith TL, Schalch H, Gaskell J, Covert S, Cullen D (1988) Nucleotide sequences of a ligninase gene from Phanerochaete chrysosporium. Nucleic Acids Res 16:1219–1219
Tien M, Kirk TK (1983) Lignin-degrading enzyme from the hymenomycete Phanerochaete chrysosporium Burds. Science 221:661–663
Tien M, Kirk TK (1984) Lignin-degrading enzyme from Phanerochaete chrysosporium: purification, characterization, and catalytic properties of a unique H2O2-requiring oxygenase. Proc Natl Acad Sci USA 81:2280–2284
Wariishi H, Akileswaran L, Gold MH (1988) Manganese peroxidase from the basidiomycete Phanerochaete chrysosporium: spectral characterization of the oxidized states and the catalytic cycle. Biochemistry 27:5365–5370
Wariishi H, Dunford HB, MacDonald ID, Gold MH (1989a) Manganese peroxidase from the lignin-degrading basidiomycete Phanerochaete chrysosporium. J Biol Chem 264:3335–3340
Wariishi H, Valli K, Renganathan V, Gold MH (1989b) Thio-mediated oxidation of nonphenolic lignin model compounds by manganese peroxidase of Phanerochaete chrysosporium. J Biol Chem 264:14 185–14 191
Wilbur WJ, Lipman DJ (1983) Rapid similarity searches of nucleic acid and protein databanks (global homology-optimal alignment). Proc Natl Acad Sci USA 80:726–730
Author information
Authors and Affiliations
Additional information
On leave from the Department of Biochemistry, University of Otago, P. O. Box 56, Dunedin, New Zealand.
Research carried out while a visiting scientist at the USDA Forest Products Laboratory from the National Chemistry Laboratory, Pune, India 41 1008
Offprint requests to: I. T. Forrester
Rights and permissions
About this article
Cite this article
Forrester, I.T., Grabski, A.C., Mishra, C. et al. Characteristics and N-terminal amino acid sequence of a manganese peroxidase purified from Lentinula edodes cultures grown on a commercial wood substrate. Appl Microbiol Biotechnol 33, 359–365 (1990). https://doi.org/10.1007/BF00164536
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00164536