Summary
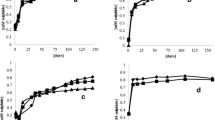
Sulphate in concentrations of 500 and 1000 mg SO4-S/l did not inhibit methanation of synthetic waste-water (acetate + methanol + glucose) by sludge from a digester treating neutral spent sulphite process effluents. The role of sulphate reducers in the conversion of those substrates was minor although sulphate-reducing bacteria were present with a viable count similar to that of methane-producing bacteria in the sludge. Neutral spent sulphite liquor was partially converted to methane (40% of chemical oxygen demand) under these conditions.
Molybdate (20 mM) inhibited methanation of both synthetic waste-water and neutral spent sulphite liquor. Acetate accumulated in glucose plus molybdate media. Molybdate had a direct inhibitory effect on enriched acetoclastic methane-producing bacteria. Molybdate was bacteriocidic to sulphate-reducing bacteria and bacteriostatic to methane-producing bacteria.
Similar content being viewed by others
References
Abram JW, Nedwell DB (1978) Inhibition of methanogenesis by sulfate-reducing bacteria competing for transferred hydrogen. Arch Microbiol 117:89–92
Badzoing W, Thauer RK, Zeikus JG (1978) Isolation and characterization ofDesulfovibrio growing on hydrogen plus sulfate as the sole energy source. Arch Microbiol 116:41–49
Banat IM, Nedwell DB, Talaat Balba M (1983) Stimulation of methanogenesis by slurries of salt marsh sediment after the addition of molybdate to inhibit sulfate reducing bacteria. J Gen Microbiol 129:123–129
Benjamin MM, Woods SL, Ferguson JF (1984) Anaerobic toxicity and biodegradability of pulp mill waste constituents. Water Res 18:601–607
Braun M, Stolp H (1985) Degradation of methanol by a sulfate reducing bacterium. Arch Microbiol 142:77–80
Bryant MP, Tzeng SF, Robinson JM, Joyner AE (1971) Nutrient requirements in methanogenic bacteria. Adv Chem Ser 105:23–40
Daniels L, Belay N, Rajagopel BS (1986) Assimilatory reduction of sulfate and sulfite by methanogenic bacteria. Appl Environ Microbiol 51:703–709
Eis BJ, Ferguson JF, Benjamin MM (1983) The fate and effect of bisulfite in anaerobic treatment. J Water Pollut Control Fed 55:1355–1365
Ferry JG, Smith PH, Wolfe RS (1974)Methanospirillum, a new genus of methanogenic bacteria, and characterization ofMethanospirillum hungatii sp. nov. Int J Syst Bacteriol 24:465–469
Frostell B (1984) Anaerobic-aerobic pilot scale treatment of a sulphite evaporator condensate. Pulp Paper Can 85:80–87
Gerhard P (1982) Diluents and biomass measurement. In: Gerhard P, Murray RGE, Costilow RN, Nester EW, Wood WA, Krieg NR, Phillips GB (eds) Manual of methods for general bacteriology. American Society for Microbiology, Washington, DC, pp 504–507
Hilton MG, Archer DB (1988) Anaerobic digestion of a sulfate-rich molasses wastewater: inhibition of hydrogen sulfide production. Biotechnol Bioeng 31:885–888
Hungate RE (1969) A roll tube method for cultivation of strict anaerobes. In: Norris JR and Ribbons DW (eds) Methods in microbiology, vol 3B. Academic Press London and New York, pp 117–132
Karhadkar PP, Audig J-M, Faup GM, Khanna P (1987) Sulfide and sulfate inhibition of methanogenesis. Water Res 21:1061–1066
Lapage SP, Shelton JE, Mitchell TG (1970) Media for the maintenance and preservation of bacteria. In: Norris JR and Ribbons DW (eds) Methods in microbiology, vol 3A, Academic Press London and New York, p 120
Lowley DR, Klug MJ (1983) Sulfate reducers can outcompete methanogens at freshwater sulfate concentrations. Appl Environ Microbiol 45:187–192
Mah RA, Smith MR (1981) The methanogenic bacteria, In: Starr MP, Stolp H, Trüper HG, Balows A, Schlegel HG (eds) The prokaryotes. A handbook on habitats, isolation, and identification of bacteria. Springer, Berlin, Heidelberg, New York, pp 948–977
Newport PJ, Nedwell DB (1988) The mechanisms of inhibition ofDesulfovibrio andDesulfotomaculum species by selenate and molybdate. J Appl Bacteriol 65:419–423
Nottingham PM, Hungate RE (1969) Methane fermentation of benzoate. J Bacteriol 98:1170–1172
Oremland RS, Polcin S (1982) Methanogenesis and sulfate reduction: competitive and noncompetitive substrates in estuarine sediments. Appl Environ Microbiol 44:1270–1276
Oremland RS, Taylor BF (1978) Sulfate reduction and methanogenesis in marine sediments. Geochim Cosmochim Acta 42:209–214
Owen WF, Stuckey DC, Healy JB Jr, Young LY, McCarty PL (1979) Bioassay for monitoring biochemical methane potential and anaerobe toxicity. Water Res 13:485–492
Peck HD Jr (1959) The ATP-dependent reduction of sulfate with hydrogen in extracts ofDesulfovibrio desulfuricans. Proc Natl Acad Sci USA 45:701–708
Puhakka JA, Ferguson JF, Benjamin MM, Salkinoja-Salonen M (1989) Sulfur reduction and inhibition in anaerobic treatment of simulated pulp mill wastewater. Syst Appl Microbiol 11:202–206
Rönnow PH, Gunnarson LAH (1981) Sulfide dependent methane production and growth of a thermophilic methanogenic bacterium. Appl Environ Microbiol 42:580–584
Schönheit P, Kristjansson JK, Thauer RK (1982) Kinetic mechanism for the ability of sulfate reducers to outcompete methanogens for acetate. Arch Microbiol 132:285–288
Standard methods for the examination of water and wastewater (1985) American Public Health Association, American Water Works Association, Water Pollution Control Federation, Washington, DC
Taylor BF, Oremland RS (1979) Depletion of adenosine triphosphate inDesulfovibrio by oxyanions of group VI elements. Curr Microbiol 42:101–103
Winfrey MR, Zeikus JG (1977) Effect of sulfate on carbon and electron flow during microbial methanogenesis in freshwater sediments. Appl Environ Microbiol 33:275–281
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Puhakka, J.A., Salkinoja-Salonen, M., Ferguson, J.F. et al. Effect of molybdate ions on methanation of simulated and natural waste-waters. Appl Microbiol Biotechnol 32, 494–498 (1990). https://doi.org/10.1007/BF00903790
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00903790