Abstract
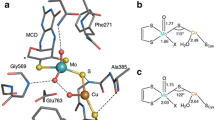
1H-NMR and electronic spectroscopic data are reported for the interaction of the effector molecule imidazole and the inhibitor molecule pyrazole with horse liver alcohol dehydrogenase whose catalytic zinc ions were replaced by Co(II). In addition 13C-NMR and optical data are given for the binding of acetate to this enzyme species. For the binary complex with imidazole an assignment of the protons of the metal-coordinated imidazole has been made and it was found that the rate of exchange of the effector molecule is slow on the NMR time scale. In the presence of NADH which is bound to the open conformation of the binary complex, the most pronounced change is a shift of the β-CH2 protons of the metal-coordinated cysteine residues which is attributed to hydrogen bonding interactions between the carboxamide group of the nicotinamide moiety with cysteine 46. The 1H-NMR spectra of the binary complex of Co(II)-HLADH with pyrazole show resonances assigned to the protons in the 3-and 4-positions of the bound inhibitor, the NH proton resonance is not detectable. In the ternary complex with pyrazole and NAD+ only the resonances of the β-CH2 protons (beyond 150 ppm) are changed whereas the protons of histidine 67 and the bound inhibitor are unchanged. The data demonstrate that the coordination environment of the catalytic metal ion is changed very little when the protein changes from the open to the closed conformation. The only changes observed are the β-CH2 proton resonances of the metal-coordinating cysteines which are sensitive to local conformational changes within the ternary complex Co(II)-HLADH · Imidazole · NADH in the open conformation or global changes in the ternary complex Co(II)-HLADH · Pyrazole · NAD+ in the closed conformation. Acetate which can be regarded as a substrate model was shown to induce a similar change in the optical spectra of the Co(II) enzyme as all other anions observed so far. From the optical changes a dissociation constant of acetate at the catalytic metal site of 200±50 mM was calculated and from the changes of the 13C-NMR linewidth of 13C acetate direct bonding of the anion to the catalytic Co(II) ion can be demonstrated to occur under the conditions of rapid exchange. The implications of these data for the assessment of tetracoordination around the catalytic metal ion as well as the chemical nature of intermediates occurring along the catalytic pathway are discussed.
Similar content being viewed by others
References
Andersson I, Zeppezauer M, Bull Th, Einarsson R, Norne J-E, Lindman B (1979a) Binding of coenzyme, coenzyme fragments, and inhibitors to native and carboxymethylated horse liver alcohol dehydrogenase from chlorine-35 nuclear magnetic resonance quadrupole relaxation. Biochemistry 18:3407–3413
Andersson I, Burton DR, Dietrich H, Maret W, Zeppezauer M (1979b) Active-site specifically reconstituted cobalt(II) horse liver alcohol dehydrogenase: optical and NMR studies on substrate, coenzyme and inhibitor binding. In: Weser U (ed) Metalloproteins. Georg Thieme Verlag, Stuttgart New York, pp 246–253
Andersson P, Kvassman J, Lindström A, Oldén B, Pettersson G (1981) Effect of pH on Pyrazole binding to liver alcohol dehydrogenase. Eur J Biochem 114:549–554
Becker NN, Roberts JD (1984) Structure of the liver alcohol dehydrogenase-NAD+-pyrazole complex as determined by 15N-NMR spectroscopy. Biochemistry 23:3336–3340
Bertini I, Luchinat C (1984) High spin cobalt (II) as probe for the investigation of metalloproteins. Adv Inorg Biochem 6: 71–111
Bertini I, Gerber M, Lanini G, Maret W, Rawer S, Zeppezauer M (1984) 1H-NMR investigation of the active site of cobalt(II)-substituted liver alcohol dehydrogenase. J Am Chem Soc 106:1826–1830
Boiwe T, Brändén C-I (1977) X-ray investigation of the binding of 1,10-phenanthroline and imidazole to horse liver alcohol dehydrogenase. Eur J Biochem 77:173–179
Brändén C-I, Jörnvall H, Eklund H, Furugren B (1975) Alcohol dehydrogenases. In: Boyer PD (ed) The enzymes, 3rd edn, vol 11. Academic Press, New York San Francisco London, pp 103–190
Cedergren-Zeppezauer E (1983) Crystal structure determination of reduced nicotinamide adenine dinucleotide complex with horse liver alcohol dehydrogenase maintained in its apo conformation by zinc-bound imidazole. Biochemistry 22:5761–5772
Cedergren-Zeppezauer E (1986) Coenzyme binding to three conformational states of horse liver alcohol dehydrogenase. In: Bertini I, Luchinat C, Maret W, Zeppezauer M (eds) Zine enzymes. Birkhäuser, Basel Boston, pp 393–415
Cedergren-Zeppezauer E, Andersson I, Ottonello S, Bignetti E (1985) X-ray analysis of structural changes induced by reduced nicotinamide adenine dinucleotide when bound to cysteine-46-carboxymethylated liver alcohol dehydrogenase. Biochemistry 24:4000–4010
Dahl KH, McKinley-McKee JS (1977) Affinity labeling of alcohol dehydrogenase. Chemical modification of the horse liver and the yeast enzymes with α-bromo-β(5-imidazolyl)-propionic acid and 1,3-dibromoacetone. Eur J Biochem 81:223–225
Dietrich H, Zeppezauer M (1982) Spectral evidence for three metal-linked ionization equilibria in the interaction of cobalt(II) horse liver alcohol dehydrogenase with conenzyme and substrate. J Inorg Biochem 17:227–235
Dworschack RT, Plapp BV (1977) pH, isotope, and substituent effects on the interconversion of aromatic substrates catalyzed by hydroxybutyrimidylated liver alcohol dehydrogenase. Biochemistry 16:2716–2725
Eklund H, Nordström B, Zeppezauer E, Söderlund G, Ohlsson I, Boiwe T, Söderberg B-O, Tapia O, Brändén C-I, Åkeson A (1976) Three-dimensional structure of horse liver alcohol dehydrogenase at 2.4 Å resolution. J Mol Biol 102:27–59
Eklund H, Samama J-P, Wallén L (1982) Pyrazole binding in crystalline binary and ternary complexes with liver alcohol dehydrogenase. Biochemistry 21:4858–4866
Gerber M, Zeppezauer M, Dunn M (1983) Evidence for innersphere coordinated alkoxide ion intermediates in the catalytic mechanism of Co(II)-substituted liver alcohol dehydrogenase. Inorg Chim Acta 79:161–164
Makinen MW, Yim MB (1981) Coordination environment of the active-site metal ion of liver alcohol dehydrogenase. Proc Natl Acad Sci USA 78:6221–6225
Makinen MW, Maret W, Yim MB (1983) Neutral metalbound water is the base catalyst in liver alcohol dehydrogenase. Proc Natl Acad Sci USA 80:2584–2588
Maret W (1980) Metalloalkoholdehydrogenasen. Methodik des Metallaustauschs, Präparation und Charakterisierung der blauen Kobalt-und Kupferalkoholdehydrogenasen. Dissertation, Universität des Saarlandes, Saarbrücken
Maret W, Zeppezauer M (1986) Influence of anions and pH on the conformational change of horse liver alcohol dehydrogenase induced by binding of oxidized nicotinamide adenine dinucleotide: binding of chloride to the catalytic metal ion. Biochemistry 25:1584–1588
Maret W, Andersson I, Dietrich H, Schneider-Bernlöhr H, Einarsson R, Zeppezauer M (1979) Site-specific substituted cobalt(II) horse liver alcohol dehydrogenase. Preparation and characterization in solution, crystalline and immobilized state. Eur J Biochem 98:501–512
Morassi R, Bertini I, Sacconi L (1973) Five-coordination in iron(II), cobalt(II) and nickel(II) complexes. Coordin Chem Rev 11:343–402
Oldén B, Pettersson G (1982) Anion binding to liver alcohol dehydrogenase. Eur J Biochem 125:311–315
Pettersson G (1986) Ionization properties of zinc-bound ligands in alcohol dehydrogenase. In: Bertini I, Luchinat C, Maret W, Zeppezauer M (eds) Zinc enzymes, Birkhäuser, Basel Boston, pp 451–464
Reynods CH, McKinley-McKee JS (1969) Anion binding to liver alcohol dehydrogenase, studied by rate of alkylation. Eur J Biochem 10:474–478
Rosenberg RC, Root CA, Wang R-H, Cerdonio M, Gray HB (1973) Nature of the ground states of cobalt(II) and nickel(II) carboxypeptidase A. Proc Natl Acad Sci USA 70:161–163
Samama J-P, Zeppezauer E, Biellman J-F, Brändén C-I (1977) The crystal structure of complexes between horse liver alcohol dehydrogenase and the coenzyme analogs 3-iodopyridine-adenine dinucleotide and pyridine-adenine dinucleotide. Eur J Biochem 81:403–409
Sartorius C, Zeppezauer M, Dunn M (1985) Detection and characterization of intermediates in the reaction catalyzed by Co(II)-substituted horse liver alcohol dehydrogenase. Rev Port Quim 27:256–257
Sartorius C, Gerber M, Zeppezauer M, Dunn M (1987) Activesite cobalt(II)-substituted horse liver alcohol dehydrogenase: characterization of intermediates in the oxidation and reduction processes as a function of pH. Biochemistry 26:871–882
Schneider G, Eklund H, Cedergren-Zeppezauer E, Zeppezauer M (1983a) Structure of complex of the active site metal-depleted horse liver alcohol dehydrogenase and NADH. EMBO J 2:685–689
Schneider G, Eklund H, Cedergren-Zeppezauer E, Zeppezauer M (1983b) Crystal structures of the active site in specifically metal-depleted and cobalt-substituted horse liver alcohol dehydrogenase derivatives. Proc Natl Acad Sci USA 80:5289–5293
Theorell H, Chance B (1951) Kinetics of the compound of horse liver alcohol deghydrogenase and reduced diphosphopyridine nucleotide. Acta Chem Scand 5:1127–1144
Theorell H, McKinley-McKee JS (1961) Mechanism of action of liver alcohol dehydrogenase. Acta Chem Scand 15: 1811–1833
Zeppezauer M (1986) The metal environment of alcohol dehydrogenase: aspects of chemical speciation and catalytic efficiency in a biological catalyst. In: Bertini I, Luchinat C, Maret W, Zeppezauer M (eds) Zinc enzymes. Birkhäuser, Basel Boston, pp 417–434
Zeppezauer M, Andersson I, Dietrich H, Gerber M, Maret W, Schneider G, Schneider-Bernlöhr H (1984) Coordination chemistry and function of the catalytic metal ion in liver alcohol dehydrogenase. J Mol Cat 23:377–387
Author information
Authors and Affiliations
Additional information
This work has been performed with contribution of the project Projetto Strategico Biotechnologie CNR and with financial support from the Deutsche Forschungsgemeinschaft, NATO, Bundesminister für Forschung und Technologie, and the Universität des Saarlandes
Rights and permissions
About this article
Cite this article
Bertini, I., Lanini, G., Luchinat, C. et al. The influence of anions and inhibitors on the catalytic metal ion in Co(II)-substituted horse liver alcohol dehydrogenase. Eur Biophys J 14, 431–439 (1987). https://doi.org/10.1007/BF00254867
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00254867