Abstract
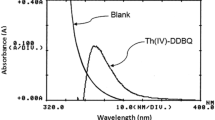
A selective and sensitive spectrophotometric method for the determination of Th(IV) has been based on the reaction with thorin and subsequent extraction of the red-orange coloured complex with N-hydroxy-N,N′-diphenylbenzamidine (HDPBA) in benzene as floated complex at pH 2.2. The complex in ethanol exhibits a maximum absorbance at 495 nm, with a molar absorptivity of 6.0 × 104 lmol−1 cm−1, with a Sandell’s sensitivity of 3.9 × 10−3 μg cm−2. The method follows Beer’s law up to 3.0 μg Th(IV)ml−1. None of the common cations and anions tested interfere. The detection limit of the method is 0.04 μg Th(IV) ml−1, the RSD (n =10) is 1.4%. The method has been successfully employed for the determination of thorium in various standard and monazite samples.
Similar content being viewed by others
References
Kavlentis E (1988) Microchem J 38: 188
Agrawal YK, Dayal U (1985) J Radioanal Nucl Chem 90: 303
Sharma SC, Tyagi MP, Purohit DN (1990) Asian J Chem 2: 214
Balon M, Munoz MA (1987) Anal Chim Acta 193: 325
Abbasi SA (1988) Anal Lett 21: 1723
Singh NSB, Thankachan TS, Balasubramanian GR (1980) J Radioanal Chem 60: 395
Tikhonov VN, Pavlova OK (1982) Zh Anal Khim 37: 1809
Valero J (1987) An Quim Ser B 83: 106
Mori I, Fujita Y, Fujita K, Koshiyama Y, Nakahashi Y (1987) Anal Lett 20: 1567
Thomason PF, Perry MA, Byerly WM (1949) Anal Chem 21: 1239
Busev AI, Tiptsova VG, Ivanov VM (1981) Analytical chemistry of rare elements, Mir Publishers, Moscow
Satyanarayana K, Mishra RK (1974) Anal Chem 46: 1609
Lange NA (1967) Handbook of chemistry, McGraw Hill, New York
Ringbom A (1932) Fresenius Z Anal Chem 115: 332
Shapiro L (1975) Rapid analysis of rock and minerals, USGS
Kirkbright GF, Johnson HN (1973) Talanta 20: 433
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nashine, N., Deb, M.K. & Mishra, R.K. Spectrophotometric determination of thorium in standard samples and monazite sands based on the floated complex of thorium with N-hydroxy-N,N′-diphenylbenzamidine and thorin. Fresenius J Anal Chem 355, 34–36 (1996). https://doi.org/10.1007/s0021663550034
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s0021663550034