Abstract
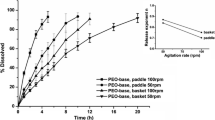
An empirical mass-transfer model for enteric-coating dissolution that uses in vitro dissolution data to characterize the pH-dependent solubility properties of the polymer film and a mass-transfer coefficient determined from in vivo dissolution or disintegration studies is developed. Once the in vivo mass- transfer coefficient has been evaluated, it can be used in conjunction with in vitro dissolution data from other formulations to predict the in vivo time to disintegration and onset of drug release. Results of in vitro dissolution experiments using the USP basket dissolution apparatus and in vivo disintegration experiments using gamma scintigraphy with four enteric-coated pellet formulations are presented. The good agreement among the in vivo mass-transfer coefficients that were determined supports the validity of the model.
Similar content being viewed by others
REFERENCES
T. Eckert, G. Cordes, and R. Seidel. Release of active substance from enteric-coated gelatine capsules in vivo and in vitro. 4. Studies with the pH endo-radio transmitter in man. Arzneim-Forsch. 21:1403–1406 (1971).
S. Rasmussen. A comparison between the in vitro and the in vivo disintegration times of enteric-coated tablets. Deut. Apoth-Ztg. 107:1321–1323 (1967).
N. Kanima, H. Ogata, N. Aoyagi, M. Koibuchi, T. Shibazaki, A. Ejima, S. Takanashi, H. Kamiyana, H. Suzuki, Y. Hinohara, H. Nakano, A. Okazaki, T. Fugikura, K. Igusa, and S. Bessho. Bioavailability of pyridoxal phosphate from enteric-coated tablets. III. Correlations between bioavailability in humans and beagel dogs and between bioavailability in humans and in vitro dissolution rates. Chem. Pharm. Bull. 33:3906–3914 (1985).
L. Borgstrom, B. Ekman, H. Larsson, I. Leden, A. Lindahl, A. Melander, and E. Wahlin-Boll. In vitro and in vivo evaluation of controlled-release and enteric-coated formulations of sodium salicylate. Biopharm. Drug Dispos. 5:261–272 (1984).
S. S. Ozturk, B. O. Palsson, B. Donohoe, and J. B. Dressman. Kinetics of release from enteric-coated tablets. Pharm. Res. 5:550–565 (1988).
R. B. Bird, W. E. Stewart, and E. N. Lightfoot. Transport Phenomena, John Wiley & Sons, New York, 1960, pp. 642–652.
D. F. Evans, G. Pye, R. Bramley, A. G. Clark, T. J. Dyson, and J. D. Hardcastle. Measurement of gastrointestinal pH profiles in normal ambulant human subjects. Gut 29:1035–1041 (1988).
H. Akima. A new method of interpolation and smooth curve fitting based on local procedures. J. ACM 17:589–602 (1970).
R. Piessens, E. deDoncker-Kapenga, C. W. Uberhuber, and D. K. Kahaner. Quadpack, Springer-Verlag, New York, 1983.
A. H. Free, E. C. Adams, M. L. Kercher, H. M. Free, and M. H. Cook. Simple specific test for urine glucose. Clin. Chem. 3:163–168 (1957).
A. Parr, M. Jay, G. A. Digenis, and R. M. Beihn. Radiolabeling of intact tablets by neutron activation for in vivo scintigraphic studies. J. Pharm. Sci. 74:590–591 (1985).
G. A. Digenis, E. P. Sandefer, A. F. Parr, R. M. Beihn, C. McClain, B. M. Scheinthal, I. Ghebre-Sellassie, U. Iyer, R. U. Nesbitt, and E. Randinitis. Gastrointestinal behavior of orally administered radiolabeled erythromycin pellets in man as determined by gamma scintigraphy. J. Clin. Pharmacol. 30:621–631 (1990).
S. S. Davis, J. G. Hardy, and J. W. Fara. Transit of pharmaceutical dosage forms through the small intestine. Gut 27:886–892 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ebel, J.P., Jay, M. & Beihn, R.M. An in Vitro/in Vivo Correlation for the Disintegration and Onset of Drug Release from Enteric-Coated Pellets. Pharm Res 10, 233–238 (1993). https://doi.org/10.1023/A:1018986827350
Issue Date:
DOI: https://doi.org/10.1023/A:1018986827350