Abstract
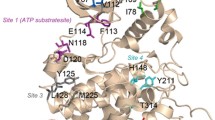
Protein kinase CK2 is characterized by a number of features, including substrate specificity, inhibition by polyanionic compounds and intrasteric down-regulation by its β-subunit, which denote a special aptitude to interact with negatively charged ligands. This situation may reflect the presence in CK2 catalytic subunits of several basic residues that are not conserved in the majority of other protein kinases. Some of these residues, notably K49 in the ‘Gly rich loop’, K74, K75, K76, K77, K79, R80, K83 in the ‘Lys rich segment’ and R191, R195, K198 in the ‘p+1 loop’, have been shown by mutational studies to be implicated to various extents and with distinct roles in substrate recognition, inhibition by heparin and by pseudosubstrate and instrasteric regulation. Molecular modelization based on crystallographic data provide a rationale for the biochemical observations, showing that several of these basic residues are clustered around the active site where they make contact with individual acidic residues of the peptide substrate. They can also mediate the effect of polyanionic inhibitors (e.g. heparin) and of regulatory elements present in the b-subunit, in the N terminal segment of the catalytic subunit and possibly in other proteins interacting with CK2. Our data also disclose a unique mode of binding of the phosphoacceptor substrate which bridges across the catalytic cleft making contacts with both the lower and upper lobes of CK2.
Similar content being viewed by others
References
Pinna LA: Casein kinase 2: An ‘eminence grise’ in cellular regulation. Biochim Biophys Acta 1054: 267–284, 1990
Tuazon PT, Traugh JA: Casein kinase I and II. Multipotential serine protein kinases: Structure, function, and regulation. In: P. Grengard, G.A. Robinson (eds). Adv Sec Mess Phos Res. Raven Press Ltd, New York, 1991, pp 123–164
Issinger O-G: Casein kinases: Pleiotropic mediators of cellular regulation. Pharmalcol Therapeut 59: 1–30, 1993
Allende JE, Allende CC: Protein kinase CK2: An enzyme with multiple substrates and a puzzling regulation. FASEB J 9: 313–323, 1995
Pinna LA, Meggio F: Protein kinase CK2 ('casein kinase-2') and its implication in cell division and proliferation. In: L. Meijer, S. Guidet, M. Philippe (eds). Progress in Cell Cycle Res. Plenum Press, New York, 1997, pp 1–21
Lasa-Benito M, Marin O, Meggio F, Pinna LA: Golgi apparatus mammary gland casein kinase: Monitoring by a specific peptide substrate and definition of specificity determinants: FEBS Lett 382: 149–152, 1996
Pinna LA, Ruzzene M: How do protein kinases recognize their substrates? Biochim Biophys Acta 1314: 191–225, 1996
Meggio F, Marin O, Pinna LA: Substrate specificity of protein kinase CK2. Cell Mol Biol Res 40: 401–409, 1994
Pinna LA, Meggio F, Marchiori F: Type 2 casein kinase: General properties and substrate specificity. In B.E. Kemp (ed). Peptides and protein phosphorylation. CRC press, 1990, pp 145–168
Sarno S, Vaglio P, Meggio F, Issinger O-G, Pinna LA: Protein kinase CK2 mutants defective in substrate recognition. Purification and kinetic analysis. J Biol Chem 271: 10595–10601, 1996
Sarno S, Vaglio P, Marin O, Issinger O-G, Ruffato K, Pinna LA: Mutational analysis of residues Implicated in the interaction between protein kinase CK2 and peptide substrates. Biochemistry 36: 11717–11724, 1997
Vaglio P, Sarno S, Marin O, Meggio F, Issinger O-G, Pinna LA: Mapping the residues of protein kinase CK2 α subunit responsible for responsiveness to polyanionic inhibitors. FEBS Lett 380: 25–28, 1996
Sarno S, Vaglio P, Marin O, Meggio F, Issinger O-G, Pinna LA: Basic residues in the 74–83 and 191–198 segments of protein kinase CK2 catalytic subunit are implicated in negative but not in positive regulation by the β-subunit. Eur J Biochem 248: 290–295, 1997
Guerra B, Niefind K, Pinna LA, Schomburg D, Issinger O-G: Expression, purification and crystallization of the catalytic subunit of protein kinase CK2 from Z. mays. Acta crystallographica D54: 143–145, 1998
Meggio F, Pinna LA: Random tyrosine and glutamic acid containing polymers are very powerful inhibitors of casein kinase-2. Biochim Biophys Acta 1010: 128–130, 1989
Miyata Y, Yahara I: Interaction between casein kinase II and the 90-kDa stress protein. HSP90. Biochemistry 34: 8123–8129, 1995
Li D, Dobrowolska G, Krebs EG: The physical association of casein kinase 2 with nucleolin. J Biol Chem 271: 15662–15668, 1996
Knighton DR, Zheng J, Ten Eyck LF, Xuong N-H, Taylor SS, Sowadski JM: Structure of a peptide inhibitor bound to the catalytic subunit of cyclic adenosine monophosphate-dependent protein kinase. Science 253: 414–420, 1991
Hubbard SR: Crystal structure of the activated insulin receptor tyrosine kinase in complex with peptide substrate and ATP analog. EMBO J 16: 5572–5581, 1997
Perich JW, Meggio F, Reynolds EC, Marin O, Pinna LA: Role of phosphorylated aminoacyl residues in generating atypical consensus sequences which are recognized by casein kinase-2 but not by casein kinase-1. Biochemistry 31: 5893–5897, 1992
Gibbs CS, Zoller MJ: Identification of electrostatic interactions that determine the phosphorylation site specificity of cAMP-dependent protein kinase. Biochemistry 30: 5329–5334, 1991
Dobrowolska G, Meggio F, Marin O, Lozeman FJ, Li D, Pinna LA, Krebs EG: Substrate recognition by casein kinase II: The role of histidine-160. FEBS Lett 355: 237–241, 1994
Niefind K, Guerra B, Pinna LA, Issinger O-G, Schomburg D: Crystal structure of the catalytic subunit of protein kinase CK2 from Zea mays 2.1 Å resolution. EMBO J 17: 2451–2462, 1998
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sarno, S., Vaglio, P., Cesaro, L. et al. A multifunctional network of basic residues confers unique properties to protein kinase CK2. Mol Cell Biochem 191, 13–19 (1999). https://doi.org/10.1023/A:1006857016712
Issue Date:
DOI: https://doi.org/10.1023/A:1006857016712