Summary
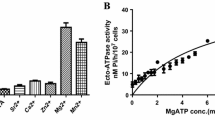
Leishmania donovani, the etiological agent for the disease visceral leishmaniasis, attach themselves to the macrophages for initiation of the disease. The attachment process has been found to be regulated by Ca2+ ions. Verapamil, a Ca2+-channel blocker inhibits Leishmania-macrophage attachment. The inhibitory effect is increased with time. Nifedipine, another Ca2+-channel blocker exhibits the same effect. The attachment process is stimulated by Ca2+-ionophore alone. The inhibitory effects of the calcium channel blockers are reversed by the ionophore.
Similar content being viewed by others
References
Benoliel AM, Capo C, Bongrand P, Rytex A, Depieds R: Non-specific binding by macrophages: existence of different adhesive mechanisms and modulation by metabolic inhibitors. Immunol 41: 547–560, 1980
Chang KP: Leishmania donovani-macrophage binding mediated by surface glycoproteins/antigens: characterization in vitro by a radioisotopic assay. Mol Biochem Parasitol 4: 67–76 1981
Chang KP: Cellular and molecular mechanisms of intracellular symbiosis in leishmaniasis. Int Rev Cytol Suppl 14: 267–302, 1983
Schatzmann HJ: ATP-Dependent Ca2+ extrusion from human red cells. Experientia 22: 364–365, 1966
Fatt P, Ginsborg BL: The ionic requirements for the production of action potentials in crustacean muscle fibres. J Physiol 142: 516–543, 1958
Gundersen RE, Nelson DL: A novel Ca+2-dependent protein kinase from Paramecium tetraaurelia. J Biol Chem 262: 4602–4609, 1987
Naitonh Y, Kaneko H: Reactivated Triton-extracted models of Paramecium: Modification of ciliary movement by calcium ions. Science 176: 523–524, 1972
Holwill MEJ, McGregor JL: Control of flagellar wave movement in Crithidia oncopelti. Nature 255: 157–158, 1975
Brokaw CJ, Josslin R, Bobrow L: Calcium ion regulation of flagellar beat symmetry in reactivated sea urchin spermatozoa. Biochem Biophys Res Commun 58: 795–800, 1974
Tash JB, Means AR: Cyclic Adenosine 3′,5′ monophosphate, calcium and protein phosphorylation in flagellar motility. Biol Reprod 28: 75–104, 1983
Eckert R, Murakami A: Calcium dependence of cilliary activity in the oviduct of the salamander necturus. J Physiol London 226: 699–711, 1972
Murakami A, Eckert R: Cilia: Activation coupled to mechanical stimulation by calcium influx. Science 175: 1375–1377, 1972
Varecka L, Carafoli E: Vanadate-induced movements of Ca2+ and K+ in human red blood cells. J Biol Chem 257: 7414–7421, 1982
Neyses L, Locher R, Stimpel M, Streuli R, Vetter W: Steriospecific modulation of the calcium channel in human erythrocytes by cholesterol and its oxidized derivatives. Biochem J 227: 105–112, 1985
Carafoli E: Intracellular calcium homeostasis. Ann Rev Biochem 56: 395–433, 1987
Channon JY, Roberts HB, Blackwell JM: A study of the differential respiratory burst activity by promastigotes and amastigotes of Leishmania donovani in murine resident peritoneal macrophages. Immunol 53: 345–355, 1984
Kalb AJ, Levitzki A: Metal binding sites of Concanavalin A and their role in the binding of α-methyl D-Glucopyranoside. Biochem J 109: 669–672, 1968
Reuter H, Seitz N: The dependence of calcium efflux from cardiac muscle on temperature and external ion composition. J Physiol 195: 451–470, 1968
Hosey MM, Lazdunski M: Calcium channels: Molecular pharmacology, structure and regulation. J Memb Biol 104: 81–105, 1988
Lux SE: Dissecting the red cell membrane skeleton. Nature (Lond) 281: 426–428, 1979
Friederichs E, Winkler H, Tillmann W: Influence of the red blood cell Ca2+ ion concentration on the erythrocyte aggregation in stasis. Biochem Med Met Biol 41: 85–92, 1989
Blackwell JM, Ezekowitz RA, Roberts MB, Channon JY, Sim RB, Gordon S: Macrophage complement and lectinlike receptors bind Leishmania in the absence of serum. J Exp Med 162: 324–331, 1985
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Misra, S., Naskar, K., Sarkar, D. et al. Role of Ca2+ ion on Leishmania-macrophage attachment. Mol Cell Biochem 102, 13–18 (1991). https://doi.org/10.1007/BF00232154
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00232154