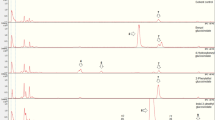
Abstract
The foliage of sweetbay magnolia (Magnolia virginana) contains at least two biologically active phenylpropanoid compounds (magnolol and a biphenyl ether) that are toxic to a number of generalist insect herbivores. These compounds have little effect on caterpillars of the sweetbay silkmoth, C. securifera, which is a specialist on sweetbay, but they are toxic to two closely related silkmoths, C. angulifera and C. promethea. To understand the influence of phytochemistry on the evolution of host use and feeding specialization in Callosamia, the detoxification capability of C. securifera was compared with that of C. angulifera and C. promethea. Degradation of magnolol and the biphenyl ether by midgut homogenate of the sweetbay specialist was NADPH-dependent and inhibited by piperonyl butoxide, suggesting the involvement of cytochrome P-450 detoxification enzymes. Both were degraded three times faster in the specialist compared to the unadapted herbivores. Higher rates of degradation could not be induced in the polyphagous C. promethea by a mixture of magnolol and the biphenyl ether or by the P-450 inducer pentamethylbenzene, nor did activity vary significantly when larvae were reared on different host plants. Use of sweetbay by Callosamia silkmoths appears to be dependent on their ability to degrade host toxins rapidly via midgut detoxification enzymes. Moreover, the intraspecific comparisons contradict the common prediction that higher levels of cytochrome P-450 activity are found in more polyphagous species; instead, P-450 activity is more closely associated with specific chemical attributes of the herbivores' host plants.
Similar content being viewed by others
REFERENCES
Ahmad, S. 1979. The functional roles of cytochrome P-450 mediated systems: Present knowledge and future areas of investigation. Drug Metab. Rev. 10:1-14.
Ahmad, S. 1983. Mixed-function oxidase activity in a generalist herbivore in relation to its biology, food plants, and feeding history. Ecology 64:235-243.
Ahmad, S., Brattsten, L. B., Mullin, C. A., and Yu, S. J. 1986. Enzymes involved in the metabolism of plant allelochemicals, pp. 73-151, in L. B. Brattsen and S. Ahmad (ed.). Molecular Aspects of Insect—Plant Associations. Plenum Press, New York.
Aucoin, R. R., PhilogÈne, B. J. R., and Arnason, J. 1991. Antioxidant enzymes as biochemical defenses against phototoxin-induced oxidative stress in three species of herbivorous Lepidoptera. Arch. Insect Biochem. Physiol. 16:139-152.
Berenbaum, M. R. 1991. Comparative processing of allelochemicals in the Papilionidae (Lepidoptera). Arch. Insect Biochem. Physiol. 17:213-221.
Berenbaum, M. R., Nitao, J. K., and Zangerl, A. R. 1991. Adaptive significance of furanocoumarin diversity in Pastinaca sativa (Apiaceae). J. Chem. Ecol. 17:207-215.
Berenbaum, M. R., Cohen, M. B., and Schuler, M. A. 1992. Cytochrome P-450 monooxygenase genes in oligophagous Lepidoptera, pp. 114-124, in C. A. Mullin and J. G. Scott (eds.). ACS Symposium Series 505. American Chemical Society, Washington, D.C.
Bernard, C. B., Arnason, J. T., PhilogÈne, B. J. R., Lam, J., and Waddell, T. 1989. Effect of lignans and other secondary metabolites of the Asteraceae on the mono-oxygenase activity of the European corn borer. Phytochemistry 28:1373-1377.
Brattsten, L. B. 1979. Ecological significance of mixed-function oxidations. Drug Metab. Rev. 10:35-59.
Brattsten, L. B. 1992. Metabolic defenses against plant allelochemicals, pp. 176-242. in G. A. Rosenthal, and M. R. Berenbaum (eds.). Herbivores: Their Interactions with Secondary Plant Metabolites, Vol. II. Ecological and Evolutionary Processes. Academic Press, New York.
Bull, D. L., Ivie, G. W., Beier, R. C., Pryor, N. W., and Oertli, E. H. 1984. Fate of photosensitizing furanocoumarins in tolerant and sensitive insects. J. Chem. Eco. 10:893-911.
Bull, D. L. Ivie, G. W., Beier, R. C., and Pryor, N. W. 1986. In vitro metabolism of a linear furanocoumarin (8-methoxypsoralen, xanthotoxin) by mixed-function oxidases of larvae of black swallowtail butterfly and fall armyworm. J. Chem. Ecol. 12:885-892.
Carino, F., Koener, J. F., Plapp, F. W., and Feyereisen, R. 1992. Constitutive overexpression of the cytochrome P-450 gene CYP6A1 in the housefly, Musca domestica, pp. 31-40, in C. A. Mullin, and J. G. Scott (eds.). ACS Symposium Series 505. American Chemical Society, Washington, D.C.
Chang, W. S., Chang, Y. H., Lu, F. I., and Chiang, H. C. 1994. Inhibitory effects of phenolics on xanthine oxidase. Anticancer Res. 14(2a):501-506.
Clark, A. M., El-Feraly, F. S., and Li, W. 1981. Antimicrobial activity of phenolic constituents of Magnolia grandiflora L. J. Pharm. Sci. 70(8):951-952.
Cohen, M. B., Berenbaum, M. R., and Schuler, M. A. 1989. Induction of cytochrome P-450-mediated detoxification of xanthotoxin in the black swallowtail. J. Chem. Ecol. 15:2347-2355.
Cohen, M. B., Schuler, M. A., and Berenbaum, M. R. 1992. A host plant inducible cytochrome P-450 from a host-specific caterpillar: Molecular cloning and evolution. Proc. Natl. Acad. Sci. U.S.A. 89:10920-10924.
Dowd, P. F., Smith, C. M., and Sparks, T. C. 1983. Detoxification of plant toxins by insects. Insect Biochem. 13:453-468.
Ehrlich, P. R., and Raven, P. R. 1964. Butterflies and plants: A study in coevolution. Evolution 18:586-608.
Faure, M., Lissi, E., Torres, R., and Videla, L. 1990. Antioxidant activities of lignans and flavonoids. Phytochemistry 29(12):3773-3775.
Ferguson, D. C. 1972. Bombycoidea, Saturniidae (in part). In R. B. Dominick (ed.). The Moths of America North of Mexico. Fascicle 20.2. Classey, Ltd. and RBD Publications, Inc., London.
Feyereisen, R., Koener, J. F., Farnsworth, D. E., and Nebert, D. W. 1989. Isolation and sequence of cDNA encoding a cytochrome P-450 from an insecticide-resistant strain of the house fly, Musca domestica. Proc. Natl. Acad. Sci. U.S.A. 86:1465-1469.
Fugita, S., and Taira, J. 1994. Biphenyl compounds are hydroxyl radical scavengers: their effective inhibition for UV-induced mutation in Salmonella typhimurium TA102. Free Radic. Biol. Med. 17(3):273-277.
Fukuyama, Y., Otoshi, Y., Miyoshi, K., Nakamura, K., Kodama, M., Nagasawa, M., Hasegawa, T., Okazaki, H., and Sugawara, M. 1992. Neurotrophic sesquiterpene neolignans from Magnolia obovata: Structure and neurotrophic activity. Tetrahedron 48(3):377-392.
Futuyma, D., and Moreno, G. 1988. The evolution of ecological specialization. Annu. Rev. Ecol. Syst. 19:207-233.
Gunderson, C. A., Brattsen, L. B., and Fleming, J. T., 1986. Microsomal oxidase and glutathione transferase as factors influencing the effects of pulegone in southern and fall armyworm larvae. Pestic. Biochem. Physiol. 26:238-249.
Haraguchi, H., Ishikawa, H., Shirataki, N., and Fukuda, A. 1997. Antiperoxidative activity of neolignans from Magnolia obovata. J. Pharm. Pharmacol. 49(2):209-212.
Hong, C. Y., Huang, S. S., and Tsai, S. K. 1996. Magnolol reduces infarct size and suppresses ventricular arrhythmia in rats subjected to coronary ligation. Clin. Exp. Pharm. Physiol. 23(8):660-664.
Johnson, K. S., Scriber, J. M., and Nair, M. 1996a. Phenylpropanoid phenolics in sweetbay magnolia as chemical determinants of host use in saturniid silkmoths (Callosamia). J. Chem. Ecol. 22:1955-1969.
Johnson, K. S., Snider, D., and Scriber, J. M. 1996b. Estimates of genetic differentiation among Callosamia species and Hyalophora cecropia (Saturniidae) using allozyme electrophoresis. J. Lepid. Soc. 50(3):217-225.
Krieger, R. I., Feeny, P. P., and Wilkinson, C. F. 1971. Detoxication enzymes in the guts of caterpillars: An evolutionary answer to plant defenses? Science 172:579-581.
Lee, K., and Berenbaum, M. R. 1992. Ecological aspects of antioxidant enzymes and glutathiones-transferases in three Papilio species. Biochem. Syst. Ecol. 20:197-207.
Lindroth, R. L. 1988. Hydrolysis of phenolic glycosides by midgut β-glucosidases in Papilio glaucus subspecies. Insect Biochem. 8:789-792.
Lindroth, R. L. 1989. Biochemical detoxication: Mechanism of differential tiger swallowtail tolerance to phenolic glycosides. Oecologia 81:219-224.
Lindroth, R. L. 1991. Differential toxicity of plant allelochemicals to insects: Roles of enzymatic detoxication systems. pp. 1-33 in E. A. Bernays (ed.). Insect-Plant Interactions. CRC Press, Boca Raton, Florida.
Lo, Y. C., Teng, C. M., Chen, C. F., Chen, C. C., and Hong, C. Y. 1994. Magnolol and honokiol isolated from Magnolia officinalis protect rat heart mitochondria against lipid peroxidation. Biochem. Pharm. 47(3):549-553.
Ma, Y., Ye, J., Fukasaku, N., Hattori, M., and Namba, T. 1988. Metabolism of magnolol from magnolia cortex (IV) Enterohepatic circulation and gastrointestinal excretion of (Ring 14C) magnolol in rats. Shoyakugaku Zasshi 42:130-134.
Ma, R., Cohen, M. B., Berenbaum, M. R., and Schuler, M. A. 1994. Black swallowtail (Papilio polyxenes) alleles encode cytochrome P-450s that selectively metabolize linear furanocoumarins. Arch. Biochem. Biophys. 310:332-340.
MACRae, W. D., and Towers, G. H. N. 1984. Biological activity of lignans. Phytochemistry 23:1207-1226.
Marty, M. A., and Krieger, R. I. 1984. Metabolism of uscharidin, a milkweed cardenolide, by tissue homogenates of monarch butterfly larvae, Danaus plexippus L. J. Chem. Ecol. 10:945-956.
Neal, J. J. 1987. Metabolic costs of mixed-function oxidase induction in Heliothis zea. Entomol. Exp. Appl. 43:175-180.
Nitao, J. K. 1989. Enzymatic adaptation in a specialist herbivore for feeding on furanocoumarin-containing plants. Ecology 70:629-635.
Nitao, J. K. 1995. Evolutionary stability of swallowtail adaptations to plant toxins, pp. 39-52, in J. M. Scriber, Y. Tsubaki, and R. C. Lederhouse (eds.). Swallowtail Butterflies: Their Ecology and Evolution. Scientific Publishers, Washington, D.C.
Nitao, J. K., Nair, M. G., Thorogood, D. L., Johnson, K. S., and Scriber, J. M. 1991. Bioactive neolignans from the leaves of Magnolia virginiana. Phytochemistry 30(7):2193-2195.
Nitao, J. K., Johnson, K. S., Scriber, J. M., and Nair, M. G. 1992. Magnolia virginiana neolignan compounds as chemical barriers to swallowtail butterfly host use. J. Chem. Ecol. 18(9):1661-1671.
Ogata, M., Hoshi, M., Shimotohno, K., Urano, S., and Endo, T. 1997. Antioxidant activity of magnolol, honokiol and related phenolic compounds. J. Amer. Oil Chem. Soc. 74(5):557-562.
Pacific, G. M., Vannucci, L., Bencini, C., Tusini, G., and Mosca, F. 1991. Sulphation of hydroxybiphenyls in human tissues. Xenobiotica 21:1113-1118.
Pardini, R. S., Bowen, C. A., Ahmad, S., and Blomquist, G. J. 1989. Adaptations to plant pro-oxidants in a phytophagous insect model: Enzymatic protection from oxidative stress, pp. 725-728, in Oxygen Radicals in Biology and Medicine. M. G. Simic, K. A. Taylor, J. F. Ward, and C. von Sonntagg (eds.). Plenum Press, New York.
Piegler, R. S. 1977. Hybridization of Callosamia (Saturniidae). J. Lepid. Soc. 31:23-34.
Piegler, R. S. 1980. Demonstration of reproductive isolating mechanisms in Callosamia (Saturniidae) by artificial hybridization. J. Res. Lepid. 19:72-81.
Powis, B., Moore, D. J., Wilke, T. J., and Santone, K. S. 1991. A high-performance liquid chromatography assay for measuring integrated biphenyl metabolism by intact cells: Its use with rat liver and human liver and kidney. Anal. Biochem. 167:191-198.
Rose, H. A. 1985. The relationship between feeding specialization and host plants to aldrin epoxidase activities of midgut homogenates in larval Lepidoptera. Ecol. Entomol. 10:455-467.
Sarker, S. D. 1997. Biological activity of magnolol: A review. Fitoterapia 68(1):3-8.
SAS Institute. 1985. SAS User's Guide: Statistics, Version 5 Edition. SAS Institute, Inc. Cary, North Carolina.
Stone, S. E. 1991. Foodplants of world Saturniidae. Lepid. Soc. Memoir 4. Lepidopterists' Society, Lawrence, Kansas.
Teng, C. M., Yu, S. M., Chen, C. C., Huang, Y. L., and Huang, T. F. 1990. EDRF-release and calcium ion channel blockade by magnolol, an antiplatelet agent isolated from Chinese herb Magnolia officinalis in rat thoracic aorta. Life Sci. 47(13):1153-1162.
Tsai, T. H., Lee, T. F., Chen, C. F., and Wang, L. C. H. 1995. Modulatory effects of magnolol on potassium-stimulated 5-hydroxytryptamine release from rat cortical and hippocampal slices. Neurosci. Lett. 186(1):49-52.
Tsai, T. H., Chou, C. J., and Chen, C. F. 1996. Pharmacokinetics and brain distribution of magnolol in the rat after intravenous bolus injection. J. Pharm. Pharmacol. 48:57-59.
Wadleigh, R. W., and Yu, S. J. 1988. Detoxification of isothiocyanate allelochemicals by glutathione transferase in three lepidopterous species. J. Chem. Ecol. 14:1279-1288.
Wang, J. P., Ho, T. F., Chang, L. C., and Chen, C. C. 1995. Anti-inflammatory effect of magnolol, isolated from Magnolia officinalis, on A23187-induced pleurisy in mice. J. Pharm. Pharmacol. 47(10):857-860.
Whittaker, R. H., and Feeny, P. P. 1971. Allelochemics: Chemical interactions between species. Science 171:757-770.
Wilkinson, L. 1990. Systat: The System for Statistics. SYSTAT, Inc., Evanston, Illinois.
Yu, S. J. 1987. Microsomal oxidation of allelochemicals in generalist (Spodoptera frugiperda) and semispecialist (Anticarsia gemmatalis) insect. J. Chem. Ecol. 13:423-436.
Yu, S. J., and Ing, R. T. 1984. Microsomal biphenyl hydroxylase of fall armyworm and its induction by allelochemicals and hostplant. Comp. Biochem. Physiol. 78C:145-152.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Johnson, K.S. Comparative Detoxification of Plant (Magnolia virginiana) Allelochemicals by Generalist and Specialist Saturniid Silkmoths. J Chem Ecol 25, 253–269 (1999). https://doi.org/10.1023/A:1020890628279
Issue Date:
DOI: https://doi.org/10.1023/A:1020890628279