Abstract
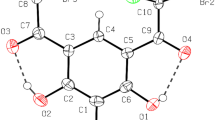
An X-ray diffraction study is reported for four molecules of mesoionic 2,3-diphenyltetrazoles. The results confirm a dipolar “mesoionic” structure, aromatic character of the tetrazole ring and no conjugation between the phenyl and tetrazole rings. The geometry of the exocyclic group is discussed in detail. The molecular parameters of the compounds investigated are correlated with13C and15N nmr data. The results obtained are compared with similar structures which have already been studied.
Similar content being viewed by others
References
Araki, S., and Butsugan, Y. (1985)Chem. Lett. 1639.
Araki, S., Miznya, J., and Butsugan, Y. (1985).J. Chem. Soc. Perkin Trans I, 2439.
Bamberger, E., Padova, R., and Ormerod, E. (1926)Annalen 446, 260.
Herbstein, F. H., Schwotzer, W. (1984)J. Chem. Soc. Perkin Trans. II 1917.
Johnson, C. K. (1976)ORTEPIIReport ORNL-5138. (Oak Ridge National Laboratory, Tenn.).
King, T. J., Preston, P. N., Suffolk, J. S., and Turnbull, K. (1979)J. Chem. Soc. Perkin Trans. II 1751.
Kozminski, W., Jazwinski, J., and Stefaniak, L. (1993)Magn. Res. Chem. In press.
Kozminski, W., Jazwinski, J., Stefaniak, L., and Webb, G. A. (1990)Magn. Res. Chem. 28, 1027.
Kushi, Y., and Fernando, Q. (1970)J. Am. Chem. Soc. 92, 1965.
Marsh, R. E., and Herbstein, F. H. (1983)Acta Cryst. B (Str. Sci.),39, 280.
Neugebauer, T. A., Fischer, H., and Krieger, C. (1979)Chem. Ber. 112, 2369.
Ollis, W. D., and Ramsden, C. A. (1976)Adv. Heterocycl. Chem. 19, 1.
Sheldrick, G. M. (1976)SHELX76. Program for crystal and molecular structure determination. (University of Cambridge, England).
Sheldrick, G. M. (1985)Crystallographic Computing 3. Sheldrick, G. M., Krüger, G., and Goddard, R. (eds.) (Oxford University Press) pp. 175–189.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Luboradzki, R., Kozminski, W. & Stefaniak, L. An X-ray diffraction study of some mesoionic 2,3-diphenyltetrazoles. Journal of Crystallographic and Spectroscopic Research 23, 133–141 (1993). https://doi.org/10.1007/BF01195447
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01195447