Abstract
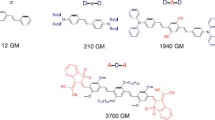
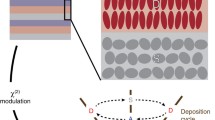
Azo-benzene chromophores with an oxygen donor and five different end-groups have been attached to octadecyl chains or to polysiloxane backbones via hexadecyl spacer groups. These were deposited by the Langmuir-Blodgett (LB) technique using our own design of diamond-barrier trough. The LB deposition properties of the polymers were superior to those of the low molecular mass materials, which did not show second harmonic generation (SHG) as deposited monolayers. The strength of SHG from the polymers correlated with acceptor strength, but the maximum signal was only about 10% of that of hemicyanine. Only one of the polymers gave thick films of very high quality; this had a hydroxyl end-group and showed no SHG except when deposited from an acid subphase. This was presumed to result from protonation of the azo group to create an acceptor. The quality of the deposited films may correlate with their phase behaviour; the best material showed only a presumed soft crystalline phase. The film-forming properties of that film were remarkable; loss figures of around 2.5 dB cm−1 have been recorded at 633 nm in films of 1.3 Μm thickness. Future work will seek to isolate and independently control the factors involved in good film formation and in high SHG.
Similar content being viewed by others
References
Y. R.Shen, The Principles of Nonlinear Optics (Wiley, New York, 1984).
R. W.Boyd, Nonlinear Optics (Academic Press, London, 1992).
G. G.Roberts, Langmuir-Blodgett Films (Plenum, New York, 1990).
A.Ulman, Ultrathin Organic Films (Academic Press, London, 1991).
D. S.Chemla and J.Zyss (eds), Nonlinear Optical Properties of Organic Molecules and Crystals (Academic Press, London, 1987).
R. H.Tredgold and M. C. J.Young, Thin Solid Films 151 (1987) 441.
M.Puterman, T.Fort and J. B.Lando, J. Colloid Interface Sci. 47 (1964) 705.
D. J.Williams, Angew. Chem. Int. Ed. Engl., 22 (1984) 690.
I. R.Girling, N. A.Cade, P. V.Kolinsky, J. D.Earls, G. H.Cross and I. r. Peterson, Thin Solid Films, 132 (1985) 101.
P. J.Travers, L. S.Miller, R. S.Sethi and M. J.Goodwin, Chemtronics 4 (1989) 239.
W.Hickel, G.Appel, D.Lupo, W.Prass and U.Scheunemann, Thin Solid Films 210 (1992) 182.
H.Finkelmann, H.Ringsdorf and J. H.Wendorff, Makromol. Chem. 177 (1978) 273.
G. W.Gray, W. D.Hawthorne, J. S.Hill, D.Lacey, M. S. K.Lee, G.Nestor and M. S.White, Polymer 30 (1989) 964.
N.Carr, M. J.Goodwin, A.McRoberts, G. W.Gray, R.Marsden and R. m. scSrowston, Makromol. Chem., Rapid Comm. 8 (1987) 487.
M. J.Goodwin, N.Carr, K.Harrison and K.Lewis, Optics Letters 16 (1991) 699.
N.Kalita, J. P.Cresswell, M. C.Petty, D.Lacey, G.Gray, M. J.Goodwin and N.Carr, Optical Materials 1 (1992) 259.
U. K. PATENT No. GB 2,209,169.
L. S.Miller, D. E.Hookes, P. J.Travers and A. P.Murphy, J. Phys. E: Sci. Instrum. 21 (1988) 163.
L. S.Miller and P. J. W.Stone, Thin Solid Films 210/211 (1992) 19.
G. J. Ashwell, E. J. C. Dawnay, A. P. Kuczynski and P. J. Martin, Physical Concepts of Materials for Novel Optoelectronic Device Applications (SPIE Proc., 1990) p. 1361.
R.Ulrich and R.Tonge, Opt. 12 (1973) 2901.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Miller, L.S., Walton, D.J., Stone, P.J.W. et al. Langmuir-Blodgett films for nonlinear optical applications. J Mater Sci: Mater Electron 5, 75–82 (1994). https://doi.org/10.1007/BF00187116
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00187116