Summary
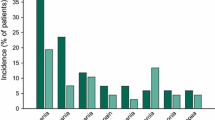
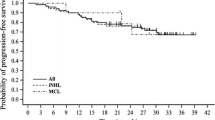
The members of the Southwest Oncology Group have treated thirteen patients with Hodgkin's disease and thirty-seven with non-Hodgkin's lymphoma with mitoxantrone on the every three week schedule. While the result (\313 responses in Hodgkin's; \937 responses in non-Hodgkin's lymphoma) is not striking, there is a definite antitumor activity in a very heavily pretreated group of patients. Toxicity was acceptable. Additional trials in lymphoma are planned using mitoxantrone in combination with BCNU.
Similar content being viewed by others
References
Von Hoff DD, Pollard E, Kuhn J, Murray E, Coltman CA Jr: Phase I clinical investigation of 1,4-dihydroxy-5,8-bis (2–2-hydroxyethyl)-amino(ethyl(amino))-9, 10-anthrace- nedione dihydrochloride (NSC 301739), a new anthracenedione. Cancer Research 40:1516- 1518, 1980
Von Hoff DD, Coltman CA Jr, Forseth B: Activity of mitoxantrone in a human tumor cloning system. Cancer Research: 41:1853–1855, 1981
Dorfman RF, Burke JS, Berard CW: A working formulation of non-Hodgkin's lymphomas: Background, recommendations, histological criteria and relationship to other classifications. In SA Rosenberg and HS Kaplan (eds): Malignant Lymphomas: Etiology, Immunology, Pathology, Treatment. Academic Press, New York 1976, pp 351–368
Dimarco A: Adriamycin (NSC-123127) mode and mechanism of action. Cancer Chemother Rep Part 3, 6:91–106, 1975
Von Hoff DD, Rozencweig M, Slavik M: Daunomycin: An anthracycline antibiotic effect in acute leukemia. Adv Pharm Chemother 15:1–50, 1978
Von Hoff DD, Rozencweig M, Lamar M, Slavik M, Muggia FM: Daunomycin-induced cardiotoxicity in children and adults. Am J Med 62:200–206, 1967
Wallace RE, Murdock KC, Augier RB, Durr FE: Activity of a novel anthracenedione, 1,3-dihydroxy-5,8-bis (2-((2-hydroxy-ethyl)amino))-9, 10-anthracenedione dihydrochloride, against experimental tumors in mice. Cancer Res 39:1570–1574, 1979
Double JC, Brown JR: Evaluation of the binding of some anthraquinones and napthacenequiones to DNA. J Pharm Pharmacol 28:116–169, 1976
Henderson BM, Dougherty WJ, James VC, Tilley LP, Noble JF: Safety assessment of a new anticancer compound, mitoxantrone, in beagle dogs: comparison with doxoru- bicin. I. Clinical Observations. Cancer Treat Rep 66:1139–1143, 1982
Sparano BM, Gordon G, Hall C, Iatropoulos MJ, Noble JF: Safety assessment of a new anticancer compound, mitoxantrone, in beagle dogs: comparison with doxorubicin. II. Histologic and ultrastructural pathology. Cancer Treat Rep 66:1145:1158, 1982
Aapro M, Mackel C, Alberts D, Woolfenden J: Phase II cardiotoxicity study of mitoxantrone hydrochloride using exercise radionuclide evaluation of the left ventricular cardiac ejection fraction (LVEF). Proc Am Soc Clin Oncol 1:14, 1982 (Abstract)
Schell FC, Yap HY, Blumenschein GR, Bodey GP: Potential cardiotoxicity observed with dihydroxyanthracenedione (DHAD) chemotherapy. Proc Am Soc Clin Oncol 1:21, 1982 (Abstract)
Unverferth DV, Balcerzak SP, Neidhart JA: Cardiac evaluation of aclacinomycin (ACLAC) and dihydroxyanthracenedione (DHAD). Proc Am Assoc Cancer Res 23:135, 1982 (Abstract)
Author information
Authors and Affiliations
Additional information
Address for reprints: Southwest Oncology Group (SWOG 8005), Operations Office, 4450 Medical Drive, San Antonio, TX 78229, U.S.A.
Rights and permissions
About this article
Cite this article
Coltman, C.A., McDaniel, T.M., Balcerzak, S.P. et al. Mitoxantrone hydrochloride (NSC-310739) in lymphoma. Invest New Drugs 1, 65–70 (1983). https://doi.org/10.1007/BF00180193
Issue Date:
DOI: https://doi.org/10.1007/BF00180193