Abstract
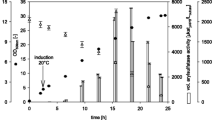
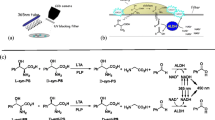
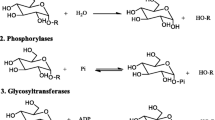
The substrate specificity of purified yeast invertase isolated fromSaccharomyces cerevisiae in transglycosylation reactions was determined. The enzyme is specific for primary alcohols. The yeast activity is a function of the alkyl length and substrate hydrophobicity (n-butyl, isobutyl, isoamyl alcohols).
Similar content being viewed by others
References
S. Kh. Abdurazakova,Improvement of Fermentation Processes Based on Stimulated Biocatalysis [in Russian], Fan, Tashkent (1990).
A. I. Oparin,Nature and Mechanism of Action of Yeast Invertase [in Russian], Academy of Sciences of the USSR, Moscow (1955).
M. M. Rakhimov,Vestn. Tashk. Gos. Univ., No. 1, 52 (1997).
D. T. Mirzarakhmetova, S. Kh.Abdurazakova, Z. R. Akhmedova, and Sh. S. Burikhanov,Khim. Prir. Soedin, 781 (1999).
Technical Ethanol, GOST 10749-79.
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 1, pp. 69–70, January–February, 2000.
Rights and permissions
About this article
Cite this article
Mirzarakhmetova, D.T., Abdurazakova, S.K., Akhmedova, Z.R. et al. Substrate specificity of yeast invertase in transglycosylation reactions. Chem Nat Compd 36, 88–89 (2000). https://doi.org/10.1007/BF02234911
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02234911