Abstract
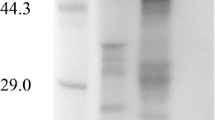
Neurospora glucamylase is a glucose-repressible extracellular enzyme. The enzyme was purified to homogeneity and found to have a molecular weight of 82,000 and to release glucose from either maltose or amylose. The rate of glucamylase synthesis increases more than 100-fold when cells are transferred from a glucose-containing medium to a glucose-free medium. Increased production of glucamylase begins within 30 min of the transfer. Glucamylase is rapidly secreted into the medium. A mutant affecting the ability of glucose to repress the synthesis of the glucose-repressible extracellular enzymes glucamylase and invertase has been isolated and studied. The mutant constitutively synthesizes and secretes a glucamylase which is indistinguishable from the wild-type enzyme.
Similar content being viewed by others
References
Carlson, M., and Botstein, D. (1982). Two differentially regulated mRNAs with different 5′ ends encode secreted and intracellular forms of yeast invertase. Cell 28145.
Carlson, M., Osmond, B. C., and Botstein, D. (1981). Mutants of yeast defective in sucrose utilization. Genetics 9825.
Davis, R. H., and DeSerres, F. J. (1970). Genetic and Microbiological research techniques for Neurospora crassa. Methods Enzymol. 1779.
Eggerding, C., Randall, J. A., and Sargent, M. L. (1975). An altered invertase in the cot-2 mutant of Neurospora crassa. J. Gen. Microbiol. 89102.
Elorza, M. V., Villanueva, J. R., and Sentandreu, R. (1977). The mechanism of catabolite inhibition of invertase by glucose in Saccharomyces cerevesiae. Biochim. Biophys. Acta 4751093.
Federoff, H. J., Eccleshall, T. R., and Marmur, J. (1983). Carbon catabolite repression of maltase synthesis in Saccharomyces carlsbergensis. J. Bacteriol. 156301.
Free, S. J., and Metzenberg, R. L. (1982). The synthesis of alkaline phosphatase in Neurospora crassa. Biochem. Genet. 20883.
Gabriel, O., and Wang, S.-F. (1969). Determination of enzymatic activity in polyacrylamide gels. I. Enzymes catalyzing the conversion of non-reducing substrates to reducing products. Anal. Biochem. 27545.
Gascon, S., and Lampen, J. O. (1968). Purification of the internal invertase of Yeast. J. Biol. Chem. 2431567.
Gratzner, H. G. (1972). Cell wall alterations associated with the hyperproduction of extracellular enzymes in Neurospora crassa. J. Bacteriol. 111443.
Hynes, M. J., and Kelly, J. M. (1977). Pleiotropic mutants of Aspergillus nidulans altered in carbon metabolism. Mol. Gen. Genet. 150193.
Laemmli, U. K. (1970). Cleavage of structural proteins during assembly of the head of bacteriophage T4. Nature 227680.
Lee, D. B., and Free, S. J. (1984). The isolation and characterization of Neurospora mutants affected in invertase synthesis. Genetics 106591.
Lineback, D. R., Russell, I. J., and Rasmussen, C. (1969). Two forms of the glucamylase of Aspergillus niger. Arch. Biochem. Biophys. 134539.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., and Randall, R. J. (1951). Protein measurement with Folin phenol reagent. J. Biol. Chem. 193265.
Metzenberg, R. L. (1979). Implications of some genetic control mechanisms in Neurospora. Microbiol. Rev. 43361.
Montecourt, B. S., Kuo, S.-C., and Lampen, J. O. (1973). Saccharomyces mutants with invertase formation resistant to repression by hexoses. J. Bacteriol. 114233.
Murayama, T., and Ishikawa, T. (1973). Mutation in Neurospora crassa affecting some of the extracellular enzymes and several growth characteristics. J. Bacteriol. 115796.
Street, H. V. (1974). Measurement of starch-iodine complex. In Bergmeyer, H. U. (ed.), Methods of Enzymatic Analysis, Vol. 2, pp. 898–903.
Takahashi, T., Inokuchi, N., and Irie, M. (1981). Purification and characterization of a glucamylase from Aspergillus saitori. J. Biochem. 89125.
Trevithick, J. R., and Metzenberg, R. L. (1966). Molecular sieving by Neurospora cell walls during secretion of invertase isozymes. J. Bacteriol. 921010.
Yamasaki, Y., Suzuki, Y., and Ozawa, J. (1977a). Purification and properties of two forms of glucamylase from Pencillium oxalicum. Agr. Biol. Chem. 41755.
Yamasaki, Y., Suzuki, Y., and Ozawa, J. (1977b). Three forms of a-glucosidase and a-glucamylase from Aspergillus awamori. Agr. Biol. Chem. 412149.
Author information
Authors and Affiliations
Additional information
Funds for this research were provided by Grant PCM-8011772 from the National Science Foundation and by a grant from the Research Development Fund of The Research Foundation of the State University of New York.
Rights and permissions
About this article
Cite this article
Sigmund, R.D., McNally, M.T., Lee, D.B. et al. Neurospora glucamylase and a mutant affected in its regulation. Biochem Genet 23, 89–103 (1985). https://doi.org/10.1007/BF00499115
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00499115