Abstract
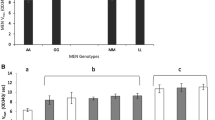
Thirteen Drosophila Adh variants have been characterized with respect to gene expression, substrate preference, thermostability, and specific activity. The results suggest that the variants may be grouped into two biochemical classes, typified by the properties of the two most common enzyme forms, ADH-F and ADH-S. Membership of these classes cannot be predicted from electrophoretic mobility, nor is any simple classification possible with regard to the characteristics of level of gene expression (in terms of ADH activity or ADH protein) or thermostability of the gene product.
Similar content being viewed by others
References
Barnes, B. W., and Birley, A. J. (1978). Genetical variation for enzyme activity in a population of Drosophila melanogaster. IV. Analysis of alcohol dehydrogenase activity in chromosome substitution lines. Heredity 4051.
Benyajati, C., Place, A R., Powers, D. A., and Sofer, W. (1981). Alcohol dehydrogenase gene of Drosophila melanogaster: Relationships of intervening sequences to functional domains in the protein. Proc. Natl. Acad. Sci. USA 782717.
Chambers, G. K. (1981). Biochemistry of alcohol dehydrogenase variation in Drosophila. In Gibson, J. B., and Oakeshott, J. G. (eds.), Genetic Studies of Drosophila Populations, Proceedings of the Kioloa Conference, Australian National University, pp. 77–93.
Chambers, G. K., McDonald, J. F., McElfresh, M., and Ayala, F. J. (1978). Alcohol oxidizing enzymes in thirteen Drosophila species. Biochem. Genet. 16757.
Chambers, G. K., Laver, W. G., Campbell, S., and Gibson, J. B. (1981). Structural analysis of an electrophoretically cryptic alcohol dehydrogenase variant from an Australian population of Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 783103.
Chambers, G. K., Wilks, A. V., and Gibson, J. B. (1981). An electrophoretically cryptic alcohol dehydrogenase variant in Drosophila melanogaster. III. Biochemical properties and comparison to the common enzyme forms Aust. J. Biol. Sci. 34625–637.
Clarke, B. (1975). The contribution of ecological genetics to evolutionary theory: Detecting the direct effects of natural selection on particular polymorphic loci. Genetics 79101.
Clarke, B., and Allendorf, F. W. (1979). Frequency-dependent selection due to kinetic differences between allozymes. Nature 279732.
David, J. (1982). Latitudinal variability of Drosophila melanogaster: Allozyme frequencies divergence between European and Afrotropical populations. Biochem. Genet. 20747.
David, J. R., De Scheemaeker-Louis, M., and Pla, E. (1980). Evolution in the seven species of the Drosophila melanogaster subgroup: Comparison of the electrophoretic mobility of the enzymes produced by the Adh locus. Dros. Inform. Serv. 5528.
Day, T. H., and Needham, L. (1974). Properties of alcohol dehydrogenase isozymes in a strain of Drosophila melanogaster homozygous for the Adh-slow allele. Biochem. Genet. 11167.
Day, T. H., Hillier, P. C., and Clarke, B. C. (1974a). Properties of genetically polymorphic isozymes of alcohol dehydrogenase in Drosophila melanogaster. Biochem. Genet. 11141.
Day, T. H., Hillier, P. C., and Clarke, B. C. (1974b). The relative quantities and catalytic activities of enzymes produced by alleles at the alcohol dehydrogenase locus in Drosophila melanogaster. Biochem. Genet 11155.
Fletcher, T. S., Ayala, F. J., Thatcher, D. R., and Chambers, G. K. (1978). Structural analysis of the ADHS electromorph of Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 755609.
Fuerst, P. A., and Ferrell, R. E. (1980). The stepwise mutation model: An experimental evaluation utilizing hemoglobin variants. Genetics 94185.
Gibson, J. B., Chambers, G. K., Wilks, A. V., and Oakeshott, J. G. (1980). An electrophoretically cryptic alcohol dehydrogenase variant in Drosophila melanogaster. I. Activity ratio, genetic localization and comparison with two other thermostable variants. Aust. J. Biol. Sci. 33479.
Gibson, J. B., Wilks, A. V., and Chambers, G. K. (1982). Genetic variation at the alcohol dehydrogenase locus in Drosophila melanogaster: A third ubiquitous allele. Experientia 38653.
Grell, E. H., Jacobson, K. B., and Murphy, J. B. (1968). Alterations of genetic material for analysis of alcohol dehydrogenase isozymes of Drosophila melanogaster. Ann. N.Y. Acad. Sci. 151441.
Johnson, F. M., and Powell, A. (1974). The alcohol dehydrogenases of Drosophila melanogaster: Frequency changes associated with heat and cold shock. Proc. Natl. Acad. Sci. USA 711783.
Johnson, G. B. (1976). Genetic polymorphism and enzyme function. In Ayala, F. J. (ed.), Molecular Evolution Sinauer, Sunderland, Mass. pp. 46–59.
Kimura, M. (1982). The neutral theory as a basis for understanding the mechanism of evolution and variation at the molecular level. In Kimura, M. (ed.), Molecular Evolution, Protein Polymorphism and the Neutral Theory Japan Scientific Societies Press, Tokyo/Springer-Verlag, Berlin, pp. 3–56.
Kimura, M., and Crow, J. V. (1964). The number of alleles that can be maintained in a finite population. Genetics 49725.
Latter, B. D. H. (1980). Gene frequency distributions in species of Drosophila. In Gibson, J. B., and Oakeshott, J. G. (eds.), Genetic Studies of Drosophila Populations, Proceedings of the Kioloa Conference, Australian National University, pp. 3–15.
Lewis, N., and Gibson, J. B. (1978). Enzyme protein amount variation in natural populations. Biochem. Genet. 16159.
Li, W. H. (1976). A mixed model of mutation for electrophoretic identity of proteins within and between populations. Genetics 83423.
Mancini, G., Carbonara, A. O., and Heremans, J. F. (1965). Immunochemical quantitation of antigens by single radial immunodiffusion. Immunochemistry 2235.
Maroni, G., Laurie-Ahlberg, C. C., Adams, D. A., and Wilton, A. N. (1982). Genetic variation in the expression of Adh in Drosophila melanogaster. Genetics 101431.
Marshall, D. R., and Brown, A. H. D. (1975). The charge-state model of protein polymorphism in natural populations. J. Mol. Evol. 6149.
McDonald, J. F., and Ayala, F. J. (1978). Genetic and biochemical basis of enzyme activity variation in natural populations. I. Alcohol dehydrogenase in Drosophila melanogaster. Genetics 89371.
McDonald, J. F., Chambers, G. K., David, J., and Ayala, F. J. (1977). An adaptive response due to changes in gene regulation: A study of Drosophila. Proc. Natl. Acad. Sci. USA 744562.
Oakeshott, J. G., Gibson, J. B., Anderson, P. R., and Champ, A. (1980). Opposing modes of selection on the alcohol dehydrogenase locus in Drosophila melanogaster. Aust. J. Biol. Sci. 33105.
Oakeshott, J. G., Gibson, J. B., Anderson, P. R., Knibb, W. R., Anderson, D. G., and Chambers, G. K. (1981). Alcohol dehydrogenase and glycerol-3-phosphate dehydrogenase clines in Drosophila melanogaster on three continents. Evolution 3686.
Ohta, T., and Kimura, M. (1973). A model of mutation appropriate to estimate the number of electrophoretically detectable alleles in a genetic population. Genet. Res. 22201.
Ramshaw, J. A. M., Coyne, J. A., and Lewontin, R. C. (1979). The sensitivity of gel electrophoresis as a detector of genetic variation. Genetics 931019.
Retzios, A. D., and Thatcher, D. R. (1979). Chemical basis of the electrophoretic variation observed at the alcohol dehydrogenase locus of Drosophila melanogaster. Biochimie 61701.
Sampsell, B. (1977). Isolation and genetic characterization of alcohol dehydrogenase thermostability variants occurring in natural populations of Drosophila melanogaster. Biochem. Genet. 15971.
Sampsell, B., and Steward, E. (1983). Alcohol dehydrogenase thermostability variants in Drosophila melanogaster: Comparison of activity ratios and enzyme levels (submitted for publication).
Schwartz, M. F., and Jörnvall, (1976). Structural analyses of mutant and wild-type alcohol dehydrogenases from Drosophila melanogaster. Eur. J. Biochem. 68159.
Schwartz, M., O'Donnell, J., and Sofer, W. (1979). Origin of the multiple forms of alcohol dehydrogenase from Drosophila melanogaster. Arch. Biochem. Biophys. 194365.
Singh, R. S., Hickey, D. A., and David, J. R. (1982). Genetic differentiation between geographically distant populations of Drosophila melanogaster. Genetics 101235.
Thatcher, D. R. (1977). Enzyme instability and proteolysis during the purification of an alcohol dehydrogenase from Drosophila melanogaster. Biochem. J. 163317.
Thatcher, D. R. (1980). The complete amino acid sequence of three alcohol dehydrogenase alleloenzymes (Adh N-11, AdhS and Adh UP) from the fruitfly Drosophila melanogaster. Biochem. J. 187875.
Thatcher, D. R., and Retzios, A. D. (1980). Mutations affecting the structure of Drosophila alcohol dehydrogenase. Protides Biol. Fluids 28157.
Thompson, J. N., Ashburner, M., and Woodruff, R. C. (1977). Presumptive control mutation for alcohol dehydrogenase in Drosophila melanogaster. Nature 270363.
Thörig, G. E. W., Schoone, A. A., and Scharloo, W. (1975). Variation between electrophoretically identical alleles at the alcohol dehydrogenase locus in Drosophila melanogaster. Biochem. Genet. 13721.
Vigue, C. L., Weisgram, P. A., and Rosenthal, E. (1982). Selection at the alcohol dehydrogenase locus of Drosophila melanogaster: Effects of ethanol and temperature. Biochem. Genet. 20681.
Wilks, A. V., Gibson, J. B., Oakeshott, J. G., and Chambers, G. K. (1980). An electrophoretically cryptic alcohol dehydrogenase variant in Drosophila melanogaster. II. Post-electrophoretical heat-treatment screening of natural populations. Aust. J. Biol. Sci. 33575.
Winburg, J. O., Thatcher, D. R., and McKinley-McKee, J. S. (1982a). Alcohol dehydrogenase from the fruitfly Drosophila melanogaster. Substrate specificity of the alleloenzymes AdhS and AdhUF. Biochim. Biophys. Acta 7047.
Winberg, J. O., Thatcher, D. R., and McKinley-McKee, J. S. (1982b). Alcohol dehydrogenase from the fruitfly Drosophila melanogaster. Inhibition studies of the alleloenzymes AdhS and AdhUF. Biochim. Biophys. Acta 70417.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chambers, G.K., Wilks, A.V. & Gibson, J.B. Variation in the biochemical properties of the Drosophila alcohol dehydrogenase allozymes. Biochem Genet 22, 153–168 (1984). https://doi.org/10.1007/BF00499295
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00499295