Abstract
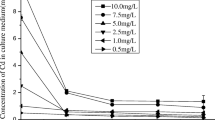
Swiss chard (Beta vulgaris L., cv. Fordhook Giant) was grown in nutrient solution with Cl concentrations varying between 0.01 mM and 120 mM. Solution Na concentration and ionic strength were maintained in all treatments by compensating with NaNO3. All solutions contained Cd (50 nM, spiked with 109Cd). Three different Cd2+ buffering systems were used. In one experiment, Cd2+ activity was unbuffered; its activity decreased with increased Cl concentration as a result of the formation of CdCln 2−n species. In the other experiments, Cd2+ activity was buffered by the chelator nitrilotriacetate (NTA, 50 μM) and ethylene-bis-(oxyethylenenitrilo)-tetraacetate (EGTA, 50 μM) at about 10−9 M and 10−11 M, respectively. Plant growth was generally unaffected by increasing Cl concentrations in the three experiments. In unbuffered solutions, Cd concentrations in plant tissue decreased significantly (p<0.01) (approximately 2.4-fold) as solution Cl concentration increased from 0.01 mM to 120 mM. However, this decrease was smaller in magnitude than the 4.7-fold decrease in Cd2+ activity as calculated by the GEOCHEM-PC program for the same range of Cl concentrations. In solutions where Cd2+ activity was buffered by NTA, Cd concentrations in plant tissue increased approximately 1.4-fold with increasing Cl concentration in solution, while the Cd2+ activity was calculated to decrease 1.3-fold. In solutions where Cd2+ activity was buffered by EGTA, Cd concentrations in the roots increased 1.3-fold with increasing Cl concentration in solution but there was no effect of Cl on shoot Cd concentrations. The data suggest that either CdCln 2−nspecies can be taken up by plant roots or that Cl enhances uptake of Cd2+ through enhanced diffusion of the uncomplexed metal to uptake sites.
Similar content being viewed by others
Abbreviations
- DAS:
-
days after sowing
- EGTA:
-
ethylene-bis-(oxyethylenenitrilo)-tetraacetate
- HBED:
-
N,N′-bis(2-hydroxybenzyl)-ethylenediamine-N,N′-diacetate
- NTA:
-
nitrilotriacetate
References
Allen H E, Hall R H and Brisbin T D 1980 Metal speciation: Effects on aquatic toxicity. Environ. Sci. Technol. 14, 441–443.
Bell P F, Chaney R L and Angle J S 1991a Determination of the free Cu2+ activity required by maize using chelator-buffered nutrient solutions. Soil Sci. Am. J. 55, 1366–1374.
Bell P F, Chaney R L and Angle J S 1991b Free metal activity and total metal concentration as indices of micronutrient availability to barley (Hordeum vulgare L. ‘Klages’). Plant and Soil 130, 51–62.
Bingham F T, Strong J E and Sposito G 1983 Influence of chloride salinity on cadmium uptake by Swiss chard. Soil Sci. 135, 160–165.
Bingham F T, Sposito G and Strong J E 1984 The effect of chloride on the availability of cadmium. J. Environ. Qual. 13, 71–74.
Cabrera D, Young S D and Rowell D L 1988 The toxicity of cadmium to barley plants as affected by complex formation with humic acid. Plant and Soil 105, 195–204.
Cataldo D A and Wildung R E 1978 Soil and plant factors influencing the accumulation of heavy metals by plants. Environ. Health Perspect. 27, 145–149.
Cataldo D A, Garland T R and Wildung R E 1983 Cadmium uptake kinetics in intact soybean plants. Plant Physiol. 73, 844–848.
Chaney R L 1988a Plants can utilize iron from Fe-N,N′-di-(2-hydroxybenzoyl)ethylenediamine-N,N′-diacetic acid, a ferric chelate with 106 greater formation constant than Fe-EDDHA. J. Plant Nutr. 11, 1033–1050.
Chaney R L 1988b Metal speciation and interaction among elements affect trace metal transfer in agricultural and environmental food chains. In Metal Speciation: Theory, Methods and Applications. Eds. RKremer and H EAllen. pp 219–260. Lewis Publishers, Inc. Chelsea, MI, USA
Checkai R T, Corey R B and Helmke P A 1987 Effects of ionic and complexed metal concentration on plant uptake of cadmium and micronutrient metals from solution. Plant and Soil 99, 335–345.
Garcia-Miragaya J and Page A L 1976 Influence of ionic strength and inorganic complex formation on sorption of trace amounts of cadmium by montmorillonite. Soil Sci. Soc. Am. J. 40, 658–663.
Giordano P M, Behel A D, Lawrence J E, Solleau J M and Bradford B N 1983 Mobility in soil and plant availability of metals derived from incinerated municipal refuse. Environ. Sci. Technol. 17, 193–198.
Hahne H C H and Kroontje W 1973 Significance of pH and chloride concentration on behaviour of heavy metal pollutants, mercury (II), cadmium (II), zinc (II) and lead (II). J. Environ. Qua. 2, 444–450.
Hatch D J, Jones L H P and Burau R G 1988 The effect of pH on the uptake of cadmium by four plant species grown in flowing solution culture. Plant and Soil 105, 121–126.
Kochian L V, Shaff J E and Lucas W J 1989 High affinity K+ uptake in maize roots. A lack of coupling with H+ efflux. Plant Physiol. 91, 1202–1211.
Laurie S H, Tancock N, McGrath S P and Sanders J R 1991 a Influence of complexation on the uptake by plants of iron, manganese, copper and zinc. I. Effect of EDTA in a multi-metal and computer simulation study. J. Exp. Bot. 42, 509–513.
Laurie S H, Tancock N, McGrath S P and Sanders J R 1991b Influence of complexation on the uptake by plants of iron, manganese, copper and zinc. II Effect of DTPA in a multi-metal and computer simulation study. J. Exp. Bot. 42, 515–519.
Li Y-M, Chaney R L and Schneiter A A 1994 Effect of soil chloride level on cadmium concentration in sunflower kernels. Plant and Soil 167, 275–280.
Lindsay W L 1979 Chemical Equilibria in Soils. John Wiley and Sons, New York, USA.
Martell A E and Smith R M 1974 Critical Stability Constants. Vol. 1, Amino Acids. Plenum Press, New York, USA.
McLaughlin M J, Tiller K G, Beech T A and Smart M K 1994 Soil salinity causes elevated cadmium concentrations in field grown potato tubers. J. Environ. Qual. 23, 1013–1018.
Parker D R, Norvell W A and Chaney R L 1995a GEOCHEM-PC: A chemical speciation model for IBM and compatible computers. In Chemical Equilibrium and Reaction Models. Eds. R HLoeppert et al. SSSA Spec. Publ. No. 42. Soil Sci. Soc. Am. Madison, WI, USA.
Parker D R, Chaney R L and Norvell W A 1995b Chemical equilibrium models: Applications to plant nutrition research. In Chemical Equilibrium and Reaction Models. Eds. R HLoeppert et al. SSSA Spec. Publ. No. 42. Soil Sci. Soc. Am. Madison, WI, USA.
Peterson C A, Emmanuel M E and Humphreys G B 1981 Pathway of movement of apoplastic fluorescent dye tracers through the endodermis at the site of secondary root formation in corn (Zea mays) and broad bean (Vicia faba). Can. J. Bot. 59, 618–625.
Römheld V and Marschner H 1981 Effect of Fe stress on utilization of Fe chelates by efficient and inefficient plant species. J. Plant Nutr. 3, 1–4.
Smolders E, McLaughlin M J and Tiller K G 1996 Influence of chloride on Cd availability to Swiss chard: a resin buffered solution culture system. Soil Sci. Soc. Am. J. (In press).
Waddington T C 1959 Lattice energies and their significance in inorganic chemistry. In Advances in Inorganic Chemistry and Radiochemistry, Eds. H JEmeléus and A GSharpe. pp 157–221. Academic Press, New-York, USA.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Smolders, E., McLaughlin, M.J. Effect of Cl on Cd uptake by Swiss chard in nutrient solutions. Plant Soil 179, 57–64 (1996). https://doi.org/10.1007/BF00011642
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00011642