Abstract
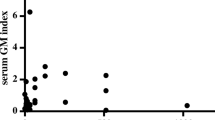
Two different kits for the detection of serum cryptococcal antigen in patients with pulmonary cryptococcosis were evaluated. The Eiken test (the Eiken Co., Tokyo), which uses pronase for pretreatment of serum, was compared with the Crypto-LA test (International Biological Laboratories, Cranbury, NJ), which did not use pronase prior to testing. Cryptococcal antigen was detected in 21 of 23 patients (91%) with the Eiken test and in only 10 of 23 patients (43%) with the Crypto-LA test (p<0.01 by Mcnemar test). However, the sensitivity of two tests was identical without use of pronase, as both tests could detect as little as 104 cells/ml ofCryptococcus neoformans and 10 ng/ml of capsular polysaccharide ofC. neoformans. In those serum specimens for which both tests were positive, titers were much higher for the Eiken test, but there was a statistically significant correlation between the two tests (coefficient correlation 0.79,p<0.01). Cryptococcal antigen titer levels measured by the Eiken test correlated well with clinical courses. There was one false-positive reaction among 82 sera of non-cryptococcal patients. Pronase enhanced the sensitivity of the Eiken test, which appeared to be useful in patients with pulmonary cryptococcal disease, and its use may prevent unneeded lung biopsies.
Similar content being viewed by others
References
Shinoda T, Ikeda R, Nishikawa A, Ohtsuka M, Futami S, Sadamoto S, Sasaki Y. Development and comparative evaluation of anti-cryptococcus neoformans antibody coated latex reagent for serodiagnosis of cryptococcosis. Jpn J Med Mycol 1989; 30: 211–21.
Small JM, Mitchell TG, Wheat R. Strain variation in composition and molecular size of the capsular polysaccharide ofCryptococcus neoformans serotype A. Infect Immun 1986; 54: 735–41.
Neill JM, Sugg JY, McCauley DW. Serologically reactive material in spinal fluid, blood and urine from a human case of cryptococcosis (torulosis). Proc Soc Exp Biol Med 1951; 77: 775–78.
Bloomfield N, Gordon MA, Elmendore DF. Detection ofCryptococcus neoformans antigen in body fluids by latex particle agglutination. Proc Soc Exp Biol Med 1963; 114: 64–67.
Kauffman CA, Germann AG, Severance PJ, McClatchey KD. Detection of cryptococcal antigen, comparison of two latex agglutination tests. Am J Clin Pathol 1981; 75: 106–9.
Wu TC, Koo S. Comparison of three commercial cryptococcal latex kits for detection of cryptococcal antigen. J Clin Microbiol 1983; 18: 1127–30.
Shinoda T, Ikeda R, Nishikawa A, Ohtsuka M, Fukazawa Y. Serodiagnosis of cryptococcosis. Jpn J Med Mycol 1985; 26: 276–83.
Jenson WA, Rose RM, Hammer SM, Karchmer AW. Serological diagnosis of focal pneumonia caused byCryptococcus neoformans. Am Rev Respir Dis 1985; 132: 189–91.
Stockman L, Roberts G. Specificity of the latex test for cryptococcal antigen: A rapid, simple method for eliminating interference factors. J Clin Microbiol 1982; 16: 965–67.
Gray LD, Roberts GD. Experience with the use of pronase to eliminate interference factors in the latex agglutination test for cryptococcal antigen. J Clin Microbiol 1988; 26: 2450–51.
McManus EJ, Bozdech MJ, Jones JM. Role of the latex agglutination test for cryptococcal antigen in diagnosing disseminated infection withTrichosporon beigelii. J Infect Dis 1985; 151: 1167–69.
Westerink MAJ, Amsterdam D, Petell RJ, Stram MN, Apicella MA. Septicemia due to DF-2. Am J Med 1987; 83: 155–58.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kohno, S., Yasuoka, A., Koga, H. et al. High detection rates of cryptococcal antigen in pulmonary cryptococcosis by Eiken Latex Agglutination test with pronase pretreatment. Mycopathologia 123, 75–79 (1993). https://doi.org/10.1007/BF01365083
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01365083