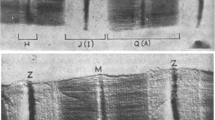
Summary
In frog semitendinosus muscle the descending limb of the length-tension curve is shifted rightward relative to that of tibialis anterior. Both the plateau right corner and the zero-force intercept are equally shifted. To investigate the reason for this shift, we compared filament lengths in the two muscles. Single fibres were mechanically skinned, stretched to reveal filaments clearly, incubated in a solution containing one of several antibodies to enhance filament visualization, and examined by electron microscopy. We found no differences of filament length. Thick filament lengths were 1.62 and 1.61 μm, respectively. I-segment lengths were measured by two methods. With the first, filament length was the same for both muscles, 1.95 or 1.98 μm, depending on the value taken for the troponin repeat; with the second it was 1.92 and 1.94 μm, respectively, for the two muscles. These differences are insignificant. Thus, the reported differences of shape of the length-tension curve are not explainable in terms of differences of filament length.
Similar content being viewed by others
References
BAGNI, M. A., CECCHI, G., COLOMO, F. & TESI, C. (1988) Plateau and descending limb of the sarcomere length-tension relation in short length-clamped segments of frog muscle fibres. J. Physiol. 401, 581–95.
BROWN, L. M., GONZALEZ-SERRATOS, H. & HUXLEY, A. F. (1984) Sarcomere and filament lengths in passive muscle fibres with wavy myofibrils. J. Muscle Res. Cell Motil. 5, 293–314.
EDMAN, K. A. P. & REGGIANI, C. (1987) The sarcomere length-tension relation determined in short segments of intact muscle fibres of the frog. J. Physiol. 385, 709–32.
FüRST, D. O., OSBORN, M., NAVE, R. & WEBER, K. (1988) The organization of titin filaments in the half sarcomere revealed by monoclonal antibodies in immunoelectron microscopy: a map of ten non-repetitive epitopes starling at the Z-line extends close to the M-line. J. Mol. Biol. 106, 1563–72.
GORDON, A. M., HUXLEY, A. F. & JULIAN, F. J. (1966a) Tension development in highly stretched vertebrate muscle fibres. J. Physiol. 184, 143–69.
GORDON, A. M., HUXLEY, A. F. & JULIAN, F. J. (1966b) The variation in isometric tension with sarcomere length in vertebrate muscle fibres. J. Physiol. 184, 170–92.
GRANZIER, H. L. M., AKSTER, H. A. & TER KEURS, H. E. D. J. (1991) Effect of thin filament length on the force-sarcomere length relation of skeletal muscle. Am. J. Physiol. (Cell) 260, 1060–70.
HANSON, J., O'BRIEN, E. J. & BENNETT, P. M. (1971) Structure of the myosin-containing filament assembly. A-segment separated from frog skeletal muscle. J. Mol. Biol. 58, 865–71.
HASELGROVE, J. C. (1975) X-ray evidence for conformational changes in the myosin filaments of vertebrate striated muscle. J. Mol. Biol. 92, 113–43.
HUXLEY, A. F. (1980) Reflections on Muscle. Liverpool, Liverpool University Press, p. 55.
IAIZZO, P. A. (1990) Histochemical and physiological properties of Rana temporaria tibialis anterior and lumbricalis IV muscle fibres. J. Muscle Res. Cell Motil. 11, 281–92.
MORGAN, D. L., CLAFLIN, D. R. & JULIAN, F. J. (1991) Tension as a function of sarcomere length and velocity of shortening in single skeletal muscle fibres of the frog. J. Physiol. 441, 719–32.
OHTSUKI, I. (1975) Distribution of troponin components in the thin filament studied by immunoelectron microscopy. J. Biochem. 77, 633–9.
OHTSUKI, I. (1979) Molecular arrangement of troponin-T in the thin filament. J. Biochem. 86, 491–7.
PAGE, S. G. (1968) Fine structure of tortoise skeletal muscle. J. Physiol. 197, 709–15.
PAGE, S. G. & HUXLEY, H. E. (1963) Filament lengths in striated muscle. J. Cell Biol. 19, 369–90.
REEDY, M. K. & REEDY, M. C. (1985) Rigor cross-bridge structure in tilted single filament layers and flared-X formations from insect flight muscle. J. Mol. Biol. 185, 145–76.
ROME, E. M., HIRABAYASHI, T. & PERRY, S. V. (1973) X-ray diffraction of muscle labelled with antibody to troponin-C. Nature New Biol. 244, 154–5.
TROMBITáS, K., BAATSEN, P. H. W. W., LIN, J. J. C., LEMANSKI, L. F. & POLLACK, G. H. (1990) Immunoelectron microscopic observations on tropomyosin localization in striated muscle. J. Muscle Res. Cell Motil. 11, 445–52.
TROMBITáS, K., BAATSEN, P. H. W. W., KELLERMAYER, M. S. Z. & POLLACK, G. H. (1991) Nature and origin of gap filaments in striated muscle. J. Cell Science 100, 809–14.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Trombitás, K., Frey, L. & Pollack, G.H. Filament lengths in frog semitendinosus and tibialis anterior muscle fibres. J Muscle Res Cell Motil 14, 167–172 (1993). https://doi.org/10.1007/BF00115451
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00115451