Abstract
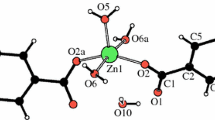
The thermal decomposition of Zn5(OH)8Cl2·H2O and β-Zn(OH)Cl materials under several experimental conditions has been studied by thermal analysis, X-ray powder diffraction and X-ray high-temperature powder diffraction techniques. Several reaction schemes are proposed to account for thermal decomposition reactions undergone by both zinc hydroxide chlorides.
Similar content being viewed by others
References
W. FEITKNECHT,Fortsch. Chem. Forschung. 2 (1953) 670.
W. NOWACKI and J. H. SILVERMAN,Z. Kristallogr. 115 (1961) 21.
A. F. WELLS, “Structural Inorganic Chemistry”, 5th Edn (Clarendon, Oxford, 1984) p. 516.
H. E. FORSBERG and W. NOWACKI,Acta Chem. Scand. 13 (1959) 1049.
B. POUSSARD and A. HARDY,Ann. Chim. Fr. 4 (1979) 403.
O. GARCÍA, E. VILA, J. L. MARTÍN de VIDALES and R. M. ROJAS,Solid State Ionics 63–65 (1993) 442.
A. MATA ARJONA, J. AYCART, O. GARCÍA and J. CANO RUIZ,Anales Quim. 63 (1967) 331.
O. GARCÍA MARTÍNEZ and J. CANO RUIZ,ibid. 63 (1967) 325.
O. K. SRIVASTAVA and E. A. SECCO,Can. J. Chem. 45 (1967) 579.
I. RASINES and J. MORALES de SETIEN,Thermochim. Acta 37 (1980) 239.
5-0664 Powder Diffraction File card.
R. C. WEAST (ed.), “Handbook of Chemistry and Physics”, 57th Edn (CRC Press, Cleveland, OH, 1976) p. B-176.
B. BREHLER,Z. Kristallogr. 115 (1961) 373.
P. RAMAMURTHY and E. A. SECCO,Can. J. Chem. 48 (1970) 2656.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Garcia-Martinez, O., Vila, E., Martin de Vidales, J.L. et al. On the thermal decomposition of the zinc(II) hydroxide chlorides Zn5(OH)8Cl2·H2O and Β-Zn(OH)Cl. J Mater Sci 29, 5429–5434 (1994). https://doi.org/10.1007/BF01171557
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01171557