Abstract
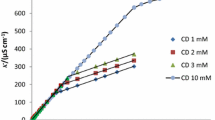
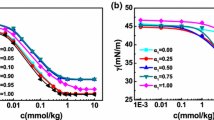
The association of anionic surfactants,viz., sodium alkyl sulfates, C n H2n+1OSO3Na (n=5, 6, 7, 8, 9, 10, 12), withβ-cyclodextrin has been investigated by means of NMR spectroscopy. The measurements of chemical shift changes of certain protons of both host and guest molecules indicated 1:1 inclusion complex formation. The association constants (K a) and standard free energy changes (−ΔG 0) for the inclusion complexation reaction vary as a function of the hydrocarbon chain length of the surfactants.
Similar content being viewed by others
References
J. Szejtli:Cyclodextrin Technology, Kluwer Academic Publishers, Dordrecht (1988).
N.J. Turro, T. Okubo and C.-J. Chung:J. Am. Chem. Soc. 104, 1789 (1982).
E. Junguera, E. Aicart and G. Tardajos:J. Phys. Chem. 96, 4533 (1992).
I. Satake, S. Yoshida, K. Hayakawa, T. Maeda and Y. Kusumoto:Bull. Chem. Soc. Jpn. 59, 3991 (1986).
T. Okubo, H. Kitano and N. Ise:J. Phys. Chem. 80, 2661 (1976).
R. Palepu and V.C. Reinsborough:Can. J. Chem. 66, 325 (1988).
J.W. Park and H.J. Song:J. Phys. Chem. 93, 6454 (1989).
J. Georgos and S. Desmettre:J. Colloid. Interface Sci. 118, 192 (1987).
E. Junquera, G. Tardajos and E. Aicart:Langmuir 9, 1213 (1993).
K.J. Sasaki, S.D. Christian and E.E. Tucker:J. Colloid. Interface Sci. 134, 412 (1990).
V.K. Smith, T. Ndou, A.M. De La Pena and I. Warner:J. Incl. Phenom. 10, 471 (1991).
R. Bergeron, M.A. Channing, G. Gibeily and D.M. Pillor:J. Am. Chem. Soc. 99, 5146 (1977).
D. Jerchel, H. Becher and M.K. Varahamutri:Z. Naturforsch. Teil 11, 681 (1956).
Z.-Z. Li, Q.-X. Guo, T. Ren, X.-Q. Zhu and Y.-C. Liu:J. Incl. Phenom. 15, 37 (1993).
H.A. Benesi and J.H. Hildebrand:J. Am. Chem. Soc. 71, 2703 (1949).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Guo, QX., Li, ZZ., Ren, T. et al. Inclusion complexation of sodium alkyl sulfates withβ-cyclodextrin. A1H NMR study. J Incl Phenom Macrocycl Chem 17, 149–156 (1994). https://doi.org/10.1007/BF00711855
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00711855