Abstract
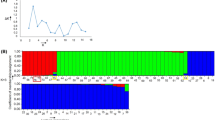
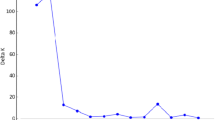
Eight Lathyrus sativus L. accessions from a variety of geographic origins were used to study intraspecific genetic diversity using RAPD analysis. Fourteen decamer primers produced 64 amplification products, 50% of which were polymorphic between the samples. Jaccard's coefficient of genetic similarity was calculated between samples and a dendrogram was constructed by an unweighted pair-group method with arithmetical averages (UPGMA). The dendrogram showed that most of the L. sativus plants clustered into accessions or common geographical areas. The average genetic similarity coefficient within accessions was 0.12 and between accessions was 0.20, indicating a low level of intraspecific genetic variation. Interspecific genetic diversity and phylogenetic relationships of eight Lathyrus species, including L. sativus and Pisum sativum L. (field pea) were examined using 14 decamer primers which produced 283 amplification products. All amplification products were polymorphic across the nine species. In the dendrogram the Lathyrus species clustered into three distinct groups which correlated with the Sections Lathyrus, Clymenum and Linearicarpus. This supports traditional taxonomic classifications of the genus Lathyrus which are based on morphological traits. Of the species from Section Lathyrus, L. gorgoni and L. cicera were the most similar to L. sativus. The results suggest that a strategy of breeding for producing lines of L. sativus with increased genetic variation would be effectively achieved through hybrid production between accessions from wide geographic areas particularly the Mediterranean area and the Indian subcontinent. However, the most effective method would be introgression of germplasm from other species in Section Lathyrus.
Similar content being viewed by others
References
Abo-elwafa, A., K. Murai & T. Shimada, 1995. Intra-and interspecific variations in Lens revealed by RAPD markers. Theor Appl Genet 90: 335–340.
Ali, S.M., B. Sharma & M.J. Ambrose, 1994. Current status and future strategy in breeding pea to improve resistance to biotic and abiotic stresses. Euphytica 73: 115–126.
Armstrong, J.S., A.J. Gibbs, R. Peakall & G. Weiller, 1994. ‘The RAPDistance Package’ Version 1.03, http://life.anu.edu.au/molecular/software/rapd.html.
Bagheri, A., J.G. Paull, P. Langridge & A.J. Rathjen, 1995. Genetic distance detected with RAPD markers among selected Australian commercial varieties and boron-tolerant exotic germplasm of pea (Pisum sativum L.). Molecular Breed 1: 193–197.
Bell, E.A., 1962. Associations of ninhydrin-reacting compounds in the seeds of 49 species of Lathyrus. Biochem J 83: 225–229.
Campbell, C.G., R.B. Mehra, S.K. Agrawal, Y.Z. Chen, A.M. Abd El Moneim, H.I.T. Khawaja, C.R. Yadov, J.U. Tay & W.A. Araya, 1994. Current status and future strategy in breeding grasspea (Lathyrus sativus). Euphytica 73: 167–175.
Chowdhury, M.A. & A.E. Slinkard, 1997. Natural outcrossing in the grasspea. J Hered 88: 154–156.
Clulow, S.A., 1989. The resistance of Pisum to Mycosphaerella pinodes (Berk. & Blox.) Vestergr. University of East Anglia (United Kingdom), PhD Thesis.
Felsenstein, J., 1985. Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39: 783–791.
Ford, R., E.C.K. Pang, & P.W.J. Taylor, 1997. Diversity analysis and species identification in Lens using PCR generated markers. Euphytica 96: 247–255.
Hoey, B.K., K.R. Crowe, V.M. Jones & N.O. Polans, 1996. A phylogenetic analysis of Pisum based on morphological characters, and allozyme and RAPD markers. Theor Appl Genet 92: 92–100.
Jackson, M.T. & A.G. Yunus, 1984. Variation in the grass pea (Lathyrus sativus L.) and wild species. Euphytica 33: 549–559.
Kearney, J.P., 1993. Wild Lathyrus species as genetic resources for improvement of grasspea (Lathyrus sativus). University of Southampton (United Kingdom), PhD Thesis.
Kupicha, F.K., 1983. The infrageneric structure of Lathyrus. Notes RBG Edinb 41: 209–244.
Link, W., C. Dixkens, M. Singh, M. Schwall & A.E. Melchinger, 1995. Genetic diversity in European and Mediterranean faba bean germ plasm revealed by RAPD markers. Theor Appl Genet 90: 27–32.
Mandal, P.K., I.M. Santha & S.L. Mehta, 1996. RAPD analysis of Lathyrus sativus somaclones. J Plant Biochem and Biotech 5: 83–86.
McComb, J.A., 1975. Is intergeneric hybridisation in the Leguminosae possible? Euphytica 24: 497–502.
Patil, P.B., P.L. Vrinten, G.J. Scoles & A.E. Slinkard, 1995. Variation in the ribosomal RNA units of the genera Lens and Cicer. Euphytica 83: 33–42.
Rahman, M.M., J. Kumar, M.A. Rahman, & M.A. Afzal, 1995. Natural outcrossing in Lathyrus sativus L. Indian J Genet 55: 204–207.
Rohlf, F.J., 1989. NTSYS-pc Version 1.80 Exeter Software, NY, USA.
Samec, P. & V. Nasinec, 1995. Detection of DNA polymorphism among pea cultivars using RAPD technique. Biologia Plantarum 37: 321–327.
Samec, P. & V. Nasinec, 1996. The use of RAPD technique for the identification and classification of Pisum sativum L. genotypes. Euphytica 89: 229–234.
Sharma, S.K., I.K. Dawson & R. Waugh, 1995. Relationships among cultivated and wild lentils revealed by RAPD analysis. Theor Appl Genet 91: 647–654.
Siddique, K.H.M., S.P. Loss, S.P. Herwig & J.M. Wilson, 1996. Growth, yield and neurotoxin (ODAP) concentration of three Lathyrus species in mediterranean-type environments of Western Australia. Aust J Expt Agric 36: 209–218.
Smartt, J., 1984. Evolution of grain legumes. I. Mediterranean pulses. Expl Agric 20: 275–296.
Taylor, P.W.J., T.A. Fraser, H.-L. Ko & R.J. Henry, 1995. RAPD analysis of sugarcane during tissue culture. In: M. Terzi, R. Cella & A. Falavigna (Eds), Current Issues in Plant Molecular and Cellular Biology, pp. 241–246. Kluwer Academic, Dordrecht Boston London.
Thormann, C.E., M.E. Ferreira, L.E.A. Camargo, J.G. Tivang & T.C. Osborn, 1994. Comparison of RFLP and RAPD markers to estimating genetic relationships within and among cruciferous species. Theor Appl Genet 88: 973–980.
Weimer, J.L., 1947. Resistance of Lathyrus spp. and Pisum spp. to Ascochyta pinodella and Mycosphaerella pinodes. J Agric Res 75: 181–190.
Williams, J.G.K., A.R. Kubelik, K.J. Livak, J.A. Rafalski & S.V. Tingey, 1990. DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucl Acids R 18: 6531–6535.
Yadav, V.K. & S.L. Mehta, 1995. Lathyrus sativus: A future pulse crop free of neurotoxin. Current Science 68: 288–292.
Yunus, A.G. & M.T. Jackson, 1991. The gene pools of the grasspea (Lathyrus sativus L.). Plant Breed 106: 319–328.
Yunus, A.G., M.T. Jackson & J.P. Catty, 1991. Phenotypic polymorphisms of six enzymes in the grasspea (Lathyrus sativus L.). Euphytica 55: 33–42.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Croft, A., Pang, E. & Taylor, P. Molecular analysis of Lathyrus sativus L. (grasspea) and related Lathyrus species. Euphytica 107, 167–176 (1999). https://doi.org/10.1023/A:1003520721375
Issue Date:
DOI: https://doi.org/10.1023/A:1003520721375