Abstract
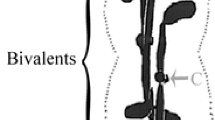
Kinetochores and microtubules were visualized simultaneously during spindle formation at the first meiotic division in microsporocytes of Lilium longiflorum (2n = 24) under a confocal laser-scanning microscope, after immunofluorescence staining with centromere-recognizing antiserum and tubulin-specific antibody. During early prometaphase I, each kinetochore of bivalent chromosomes appeared to be an amorphous flat structure upon its initial attachment to microtubules. It became compact and spherical with the development of the spindle. From late prometaphase I, when the bipolar spindle was nearly complete, each kinetochore resembled a double disk that was suggestive of a pair of sister kinetochores and the homologous kinetochores were oriented towards opposite poles. Thus, the bipolar spindle at metaphase I included 12 bivalent chromosomes with a total of four kinetochores each. At anaphase I, the sister kinetochores moved to the same spindle pole as a paired unit. In microsporocytes arrested at prometaphase I by colchicine treatment, the sister kinetochores also came to be distinguishable. These results suggest that the change of kinetochore structure during meiosis I may be under chromosomal control but be somewhat associated with its attachment to spindle microtubules.
Similar content being viewed by others
References
Braselton JP, Bowen CC (1971) The ultrastructure of the kinetochores of Lilium longiflorum during the first meiotic division. Caryologia 24: 49-58.
Brinkley BR, Brenner SL, Hall JM, Tousson A, Balczon RD, Valdivia MM (1986) Arrangements of kinetochores in mouse cells during meiosis and spermatogenesis. Chromosoma 94: 309-317.
Church K, Lin H-PP (1985) Kinetochore microtubules and chromosome movement during prometaphase in Drosophila melanogaster spermatocytes studied in life and with the electron microscope. Chromosoma 92: 273-282.
Giménez-Abián JF, Clarke DJ, García De La Vega C, Giménez-Martín G (1997) The role of sister chromatid cohesiveness and structure in meiotic behaviour. Chromosoma 106: 422-434.
Goldstein LSB (1981) Kinetochore structure and its role in chromosome orientation during the first meiotic division in male D. melanogaster. Cell 25: 591-602.
Hays TS, Salmon ED (1990) Poleward force at the kinetochore in metaphase depends on the number of kinetochore microtubules. J Cell Biol 110: 391-404.
Khodjakov A, Cole RW, McEwen BF, Buttle KF, Rieder CL (1997) Chromosome fragments possessing only one kinetochore can congress to the spindle equator. J Cell Biol 136: 229-240.
Li X, Nicklas RB (1997) Tension-sensitive kinetochore phosphorylation and the chromosome distribution checkpoint in praying mantid spermatocytes. J Cell Sci 110: 537-545.
Luykx P (1965) Kinetochore-to-pole connections during prometaphase of the meiotic divisions in urchis eggs. Exp Cell Res 39: 658-668.
Mineyuki Y, Wick SM, Gunning BES (1988) Preprophase bands of microtubules and the cell cycle: kinetics and experimental uncoupling of their formation from the nuclear cycle in onion root-tip cells. Planta 174: 518-526.
Miyazaki WY, Orr-Weaver TL (1994) Sister-chromatid cohesion in mitosis and meiosis. Annu Rev Genet 28: 167-187.
Mole-Bajer J, Bajer AS, Zinkowski RP, Balczon RD, Brinkley BR (1990) Autoantibodies from a patient with scleroderma CREST recognized kinetochores of the higher plant Haemanthus. Proc Natl Acad Sci USA 87: 3599-3603.
Nicklas RB (1997) How cells get the right chromosomes. Science 275: 632-637.
Pluta AF, Mackay AM, Ainsztein AM, Goldberg IG, Earnshaw WC (1995) The centromere: hub of chromosomal activities. Science 270: 1591-1594.
Rieder CL (1982) The formation, structure, and composition of the mammalian kinetochore and kinetochore fiber. Int Rev Cytol 79: 1-58.
Rieder CL, Palazzo RE (1992) Colcemid and the mitotic cycle. J Cell Sci 102: 387-392.
Rufas JS, Gimenez-Abian J, Suja JA, Garcia De La Vega C (1987) Chromosome organization in meiosis revealed by light microscope analysis of silver-stained cores. Genome 29: 706-712.
Schmit A-C, Stoppin V, Chevrier V, Job D, Lambert A-M (1994) Cell cycle dependent distribution of a centrosomal antigen at the perinuclear MTOC or at the kinetochores of higher plant cells. Chromosoma 103: 343-351.
Schrader F (1939) The structure of the kinetochore at meiosis. Chromosoma 1: 230-237.
Sheldon JM, Dickinson HG (1986) Pollen wall formation in Lilium: the effect of chaotropic agents, and the organization of the microtubular cytoskeleton during pattern development. Planta 168: 11-23.
Suzuki T, Ide N, Tanaka I (1997) Immunocytochemical visualization of the centromeres during male and female meiosis in Lilium longiflorum. Chromosoma 106: 435-445.
Takegami MH, Yoshioka M, Tanaka I, Ito M (1981) Characteristics of isolated microsporocytes from liliaceous plants for studies of the meiotic cell cycle in vitro. Plant Cell Physiol 22: 1-10.
Tanaka I (1991) Microtubule-determined plastid distribution during microsporogenesis in Lilium longiflorum. J Cell Sci 99: 21-31.
Traas JA, Burgain S, Dumas De Vaulx R (1989) The organization of the cytoskeleton during meiosis in eggplant (Solanum melongena (L.)): microtubules and F-actin are both necessary for coordinated meiotic division. J Cell Sci 92: 541-550.
Williams BC, Gatti M, Goldberg ML (1996) Bipolar spindle attachments affect redistributions of ZW10, a Drosophila centromere/kinetochore component required for accurate chromosome segregation. J Cell Biol 134: 1127-1140.
Wilson HJ (1968) The fine structure of the kinetochore in meiotic cells of Tradescantia. Planta 78: 379-385.
Zinkowski RP, Meyne J, Brinkley BR (1991) The centromere-kinetochore complex: a repeat subunit model. J Cell Biol 113: 1091-1110.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Suzuki, T., Tanaka, I. Attachment of kinetochores to spindle microtubules during meiosis I of Lilium microsporocytes. Chromosome Res 7, 121–130 (1999). https://doi.org/10.1023/A:1009247116452
Issue Date:
DOI: https://doi.org/10.1023/A:1009247116452