Abstract
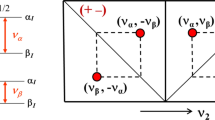
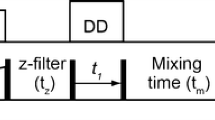
A triple-resonance NMR technique suitable for the determination ofcarbonyl-related couplings in polypeptide systems is introduced. Theapplication of three novel pulse sequences to uniformly13C/15N-enriched proteins yields E.COSY-likemultiplet patterns exhibiting either one of the3J(C′i−1,Hi α), 3J(C′i−1,Ci β) and3J(C′i−1,C′i)coupling constants in the indirectly detected 13C′dimension, depending on the passive spin selected. The experiments aredemonstrated with oxidized flavodoxin from Desulfovibrio vulgaris. On thebasis of the J-values measured and the backbone φ-angles derived from ahigh-resolution X-ray structure of the protein, the three associated Karplusequations were reparametrized. The root-mean-square differences between theexperimental coupling constants and those predicted by the optimized Karpluscurves are 0.41, 0.33 and 0.32 Hz for3J(C′i−1,Hi α),3J(C′i−1,Ci β) and3J(C′i−1,C′i),respectively. The results are compared with the Karplus parameters previouslypublished for the same couplings.
Similar content being viewed by others
References
Bax, A., Vuister, G.W., Grzesiek, S., Delaglio, F., Wang, A.C., Tschudin, R. and Zhu, G. (1994) Methods Enzymol., 239, 79–105.
Biamonti, C., Rios, C.B., Lyons, B.A. and Montelione, G.T. (1994) Adv. Biophys. Chem., 4, 51–120.
Boyd, J., Soffe, N., John, B., Plant, D. and Hurd, R. (1992) J. Magn. Reson., 98, 660–664.
Brüschweiler, R. and Case, D.A. (1994) J. Am. Chem. Soc., 116, 11199–11200.
Bystrov, V.F., Gavrilov, Yu.D. and Solkan, V.N. (1975) J. Magn. Reson., 19, 123–129.
Bystrov, V.F. (1976) Prog. NMR Spectrosc., 10, 41–81.
Case, D.A., Dyson, H.J. and Wright, P.E. (1994) Methods Enzymol., 239, 392–416.
Davis, A.L., Keeler, J., Laue, E.D. and Moskau, D. (1992) J. Magn. Reson., 98, 207–216.
DeMarco, A., Llinás, M. and Wüthrich, K. (1978a) Biopolymers, 17, 617–636.
DeMarco, A., Llinás, M. and Wüthrich, K. (1978b) Biopolymers, 17, 637–650.
DeMarco, A., Llinás, M. and Wüthrich, K. (1978c) Biopolymers, 17, 2727–2742.
DeMarco, A. and Llinás, M. (1979) Biochemistry, 18, 3846–3854.
Eberstadt, M., Gemmecker, G., Mierke, D.M. and Kessler, H. (1995) Angew. Chem., Int. Ed. Engl., 34, 1671–1695.
Emsley, L. and Bodenhausen, G. (1990) Chem. Phys. Lett., 165, 469–476.
Fischman, A.J., Live, D.H., Wyssbrod, H.R., Agosta, W.C. and Cowburn, D. (1980) J. Am. Chem. Soc., 102, 2533–2539.
Griesinger, C., Sørensen, O.W. and Ernst, R.R. (1985) J. Am. Chem. Soc., 107, 6394–6396.
Griesinger, C., Sørensen, O.W. and Ernst, R.R. (1986) J. Chem. Phys., 85, 6837–6852.
Griesinger, C., Sørensen, O.W. and Ernst, R.R. (1987) J. Magn. Reson., 75, 474–492.
Grzesiek, S. and Bax, A. (1992) J. Magn. Reson., 96, 432–440.
Grzesiek, S. and Bax, A. (1993a) J. Am. Chem. Soc., 115, 12593–12594.
Grzesiek, S. and Bax, A. (1993b) J. Biomol. NMR, 3, 627–638.
Grzesiek, S., Vuister, G.W. and Bax, A. (1993) J. Biomol. NMR, 3, 487–493.
Hansen, P.E., Feeney, J. and Roberts, G.C.K. (1975) J. Magn. Reson., 17, 249–261.
Harbison, G.J. (1993) J. Am. Chem. Soc., 115, 3026–3027.
Hoch, J.C., Dobson, C.M. and Karplus, M. (1985) Biochemistry, 24, 3831–3841.
Hosur, R.V., Ravi Kumar, M. and Sheth, A. (1985) J. Magn. Reson., 65, 375–381.
Hu, S.-J. and Bax, A. (1996a) J. Am. Chem. Soc., 118, 8170–8171.
Hu, S.-J. and Bax, A. (1996b) XVIIth International Conference on Magnetic Resonance in Biological Systems, August 18–23, 1996, Keystone, U.S.A., poster TP21.
Hu, S.-J. and Bax, A. (1997) J. Am. Chem. Soc., 119, 6360–6368.
Kao, L.-F. and Barfield, M. (1985) J. Am. Chem. Soc., 107, 2323–2330.
Karplus, M. (1959) J. Chem. Phys., 30, 11–15.
Karplus, M. (1963) J. Am. Chem. Soc., 85, 2870–2871.
Kay, L.E., Xu, G.Y. and Yamazaki, T. (1994) J. Magn. Reson., A109, 129–133.
Knauf, M., Löhr, F., Blümel, M., Mayhew, S.G. and Rüterjans, H. (1996) Eur. J. Biochem., 238, 423–434.
Krishnamurthy, V.V. (1995) J. Magn. Reson., A114, 88–91.
Li, Y.-C. and Montelione, G.T. (1993) J. Magn. Reson., B101, 315–319.
Löhr, F. and Rüterjans, H. (1995) J. Biomol. NMR, 5, 25–36.
Löhr, F. and Rüterjans, H. (1996) 13th European Experimental NMR Conference, May 19–24, 1996, Paris, France, poster p80.
Löhr, F. and Rüterjans, H. (1997) J. Am. Chem. Soc., 119, 1468–1469.
Ludvigsen, S., Andersen, K.V. and Poulsen, F.M. (1991) J. Mol. Biol., 217, 731–736.
Marion, D., Ikura, M. and Bax, A. (1989a) J. Magn. Reson., 84, 425–430.
Marion, D., Ikura, M., Tschudin, R. and Bax, A. (1989b) J. Magn. Reson., 85, 393–399.
Nagayama, K. (1986) J. Magn. Reson., 66, 240–249.
Norwood, T.J. (1993) J. Magn. Reson., A101, 109–112.
Norwood, T.J. and Jones, K. (1993) J. Magn. Reson., A104, 106–110.
Pardi, A., Billeter, M. and Wüthrich, K. (1984) J. Mol. Biol., 180, 741–751.
Piotto, M., Saudek, V. and Sklenář, V. (1992) J. Biomol. NMR, 2, 661–665.
Press, W.H., Flannery, B.P., Teukolsky, S.A. and Vetterling, W.T. (1989) Numerical Recipes, Cambridge University Press, Cambridge, U.K.
Schmidt, J.M., Ernst, R.R., Aimoto, S. and Kainosho, M. (1995) J. Biomol. NMR, 6, 95–105.
Shaka, A.J., Barker, P.B. and Freeman, R. (1985) J. Magn. Reson., 64, 547–552.
Shaka, A.J., Lee, C.J. and Pines, A. (1988) J. Magn. Reson., 77, 274–293.
Solkan, V.N. and Bystrov, V.F. (1974) Izv. Akad. Nauk SSSR, Ser. Khim., 1308–1313.
Stockman, B.J., Richardson, T.E. and Swenson, R.P. (1994) Biochemistry, 33, 15298–15308.
Stonehouse, J., Shaw, G.L., Keeler, J. and Laue, E.D. (199) J. Magn. Reson., A107, 178–184.
Szyperski, T., Braun, D., Fernández, C., Bartels, C. and Wüthrich, K. (1995) J. Magn. Reson., B108, 197–203.
Vuister, G.W. and Bax, A. (1993) J. Am. Chem. Soc., 115, 7772–7777.
Wang, A.C. and Bax, A. (1995) J. Am. Chem. Soc., 117, 1810–1813.
Wang, A.C. and Bax, A. (1996) J. Am. Chem. Soc., 118, 2483–2494.
Watenpaugh, K.D., Sieker, L.C., Jensen, L.H., LeGall, J. and Dubourdieu, M. (1972) Proc. Natl. Acad. Sci. USA, 69, 3185–3188.
Watenpaugh, K.D., Sieker, L.C. and Jensen, L.H. (1973) Proc. Natl. Acad. Sci. USA, 70, 3857–3860.
Watt, W., Tulinsky, A., Swenson, R.P. and Watenpaugh, K.D. (1991) J. Mol. Biol., 218, 195–208.
Zhu, G. and Bax, A. (1990) J. Magn. Reson., 90, 405–410.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Löhr, F., Blümel, M., Schmidt, J.M. et al. Application of H(N)CA,CO-E.COSY experiments for calibrating the φ angular dependences of vicinal couplings J(C′i−1,Hi α), J(C′i−1,Ci β) and J(C′i−1,C′i) in proteins. J Biomol NMR 10, 107–118 (1997). https://doi.org/10.1023/A:1018355327792
Issue Date:
DOI: https://doi.org/10.1023/A:1018355327792