Synopsis
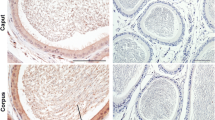
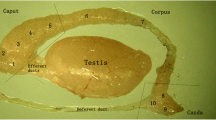
This article reports the application of Hayashi's histochemical technique for β-glucoronidase to mouse epididymis. A methodological study. which established optimal conditions for demonstrating the enzyme in this organ, is reported. The distribution pattern of β-glucuronidase is described and correlated with previous data for α-naphtyl acetate esterase. Differences between sites of granular and diffuse reaction product for these two enzymes are recorded. Possible interpretations of these findings in terms of intracellular localization of enzymes are discussed. Studies on different strains reveal regular differences in histochemical organization between mice of various genotypes. Histochemical data which imply androgen inducibility of β-glucuronidase in mouse epididymis are preliminarily noted.
Similar content being viewed by others
References
Allen, J. M. &Slater, J. J. (1957). A chemical and histochemical study of alkaline phosphatase and aliesterase in the epididymis of normal and castrate mice.Anat. Rec. 129, 255–73.
Blecher, S. R. &Kirkeby, S. (1978). Histochemical studies on genetical control of hormonal enzyme inducibility in the mouse. I. Non-specific esterase activity and regional histology of the epididymis.J. Anat. 125, 247–65.
Brandt, E. J., Elliott, R. W. &Swank, R. T. (1975). Defective lysosmal enzyme secretion in kidneys of Chediak-Higashi (beige) mice.J. Cell Biol. 67, 774–88.
Brooks, D. E. (1976). Control of glycolytic enzymes by androgens in the rat epididymis.J. Endocr. 71, 355–65.
Chi, E. Y., Ignacio, E. &Lagunoff, D. (1978). Mast cell granule formation in the beige mouse.J. Histochem. Cytochem. 26, 131–7.
Cohen, S. L. (1951). The steroidal hormones and tissae β-glucuronidase and esterase.Ann. N. Y. Acad. Sci. 54, 558–68.
Conchie, J., Findlay, J. &Levvy, G. A. (1959). Mammalian glycosidases: Distribution in the body.Biochem. J. 71, 318–25.
Dofuku, R., Tettenborn, U. &Ohno, S. (1971). Testostorone-‘regulon’ in the mouse kidney.Nature 232, 5–7.
Drews, U., Blecher, S. R., Owen, D. A. &Ohno, S. (1974). Genetically directed preferential X-activation seen in mice.Cell 1, 3–8.
Fishman, W. H. (1951). β-glucuronidase and the action of steroid hormones.Ann. N.Y. Acad. Sci. 54, 548–57.
Fishman, W. H. &Baker, J. R. (1956). Cellular localization of β-glucuronidase in rat tissues.J. Histochem. Cytochem. 4, 570–87.
Fishman, W. H., Ide, H. &Rufo, R. (1969). Dual localization of acid hydrolases in endoplasmic reticulum and in lysosomes. 1 β-Glucuronidase staining reactions and cytochemical studies on kidney in androgen-stimulated mice.Histochemie 20, 287–99.
Ganschow, R. E. &Bunker, B. G. (1970). Genetic control of glucuronidase in mice.Biochem. Genet. 4, 127–33.
Hayashi, M. (1964). Distribution of β-glucuronidase activity in rat tissues employing the naphthol AS-BI glucuronide hexazonium pararosanilin method.J. Histochem. Cytochem. 12, 659–69.
Hayashi, M. (1967). Comparative histochemical localization of lysosomal enzymes in rat tissues.J. Histochem. Cytochem. 15, 83–92.
Hayashi, M., Nakajima, Y. &Fishman, W. H. (1964). The cytologic demonstration of β-glucuronidase employing naphthol AS-BI glucuronide and hexazonium pararosanilin; a preliminary report.J. Histochem. Cytochem. 12, 293–7.
Ide, H. &Fishman, W. H. (1969). Dual localization of β-glucuronidase and acid phosphatase in lysosomes and in microsomes. II. Membrane-associated enzymes.Histochemie 20, 300–21.
Iost, A. (1961). The role of fetal hormones in prenatal development. In:The Harvey Leiures, Series 55, pp. 201–226. New York: Academic Press.
Iost, A., Vigier, B., Prépin, J. &Perchellet, J. P. (1973). Studies on sex differentiation in mammals.Recent Prog. Horm. Res. 29, 1–41.
Kato, K., Hirohata, I., Fishman, W. H. &Tsukamoto, H. (1972). Intracellular transport of mouse kidney β-glucuronidase induced by gonadotrophin.Biochem. J. 127, 425–35.
Kirkeby, S. & Blecher, S. R. (1978). Histochemical studies on genetical control of hormonal enzyme inducibility in the mouse. II. Esterase isozymes of the normal epididymis demonstrated by substrate variation and by enzyme inhibitors.Acta Histochemica et Cytochemica (in press).
Lane, P. W. &Murphy, E. D. (1972). Susceptibility to spontaneous pneumonitis in an inbred strain of beige and satin mice.Genetics 72, 451–60.
Levvy, G. A. &Marsh, C. A. (1959). Preparation and properties of β-glucuronidase.Adv. Carbohydr. Chem. 14, 381–428.
Lin, C.-W. (1975). Multiple forms of β-glucuronidase: molecular nature, transformation, and subeellular translocation. In:Isozymes (ed. C. L. Markert). pp. 637–651. London: Academic Press.
Lin, C.-W., Orcutt, M. L. &Fishman, W. H. (1975). Purification and characterization of mouse kidney β-glucuronidase.J. Biol. Chem. 250, 4737–43.
Lojda, Z. (1971). Indigogenic methods for glycosidases. IV. An improved method for β-glucuronidase.Histochemie 27, 182–92.
Milone, M. &Rastogi, R. K. (1976). Effect of testosterone upon the β-glucuronidase activity of some target tissues of the frog,Rana esculenta.J. Endocr. 70, 329–30.
Moniem, K. A. &Glover, T. D. (1972). Comparative histochemical localization of lysosomal enzymes in mammalian epididymides.J. Anat. 111, 437–52.
Oliver, C. &Essner, E. (1973). Distribution of anomalous lysosomes in the beige mouse: a homologue of Chediak-Higashi syndrome.J. Histochem. Cytochem. 21, 218–28.
Paigen, K., Swank, R. T., Tomino, S. &Ganschow, R. E. (1975). The molecular genetics of mammalian glucuronidase.J. Cell. Physiol. 85, 379–92.
Pearse, A. G. E. (1972).Histochemistry: Theoretical and Applied. Vol. 2. 3rd Edn. p. 812. Edinburgh: Churchill-Livingstone.
Pearson, B., Standen, A. C. &Esterly, J. R. (1967). Histochemical β-glucuronidase distribution in mammalian tissue as detected by 5-bromo-4-chloroindol-3-yl-β-d-glucopyruroniside.Lab. Invest. 17, 217–24.
Rutenburg, A. M., Rutenburg, S. H., Monis, B., Teague, R. &Seligman, A. M. (1958). Histochemical demonstration of β-d-galactosidase in the rat.J. Histochem. Cytochem. 6, 122–9.
Swank, R. T. &Paigen, K. (1973). Genetic control of glucuronidase induction in mice.J. Mol. Biol. 81, 225–43.
Swank, R. T., Davey, R., Joyce, L., Reid, P. &Macey, M. R. (1977). Differential effect of hypophysectomy on the synthesis of β-glucuronidase and other androgeninducible enzymes in mouse kidney.Endocrinology 100, 473–80.
Tettenborn, U., Dofuku, R. &Ohno, S. (1971). Noninducible phenotype exhibited by a proportion of female mice heterozygous for the X-linked testicular feminization mutation.Nature 234, 37–40.
Yarborough, D. J., Meyer, O. T., Dannenberg, A. M., Jr. &Pearson, B. (1967). Histochemistry of macrophage hydrolases, III. Studies on β-galactosidase, β-glucuronidase and aminopeptidase with indolyl and naphthyl substrates.J. Reticuleondothel. Soc. 4, 390–408.
Author information
Authors and Affiliations
Additional information
Present address: Department of Anatomy, Faculty of Medicine, Sir Charles Tupper Medical Building, Dalhousie University, Halifax, Nova Scotia, Canada.
Rights and permissions
About this article
Cite this article
Blecher, S.R., Kirkeby, S. Histochemical studies on genetical control of hormonal enzyme inducibility in the mouse. III. β-Glucuronidase distribution pattern of epididymis in different genotypes. Histochem J 11, 205–222 (1979). https://doi.org/10.1007/BF01002997
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01002997