Summary
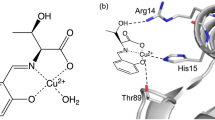
A Cu(II) complex of desferrithiocin fromStreptomyces antibioticus was prepared and characterized. The first shell atoms, including one nitrogen and four oxygens, were arranged around the copper in a square-planar pyramide. Due to the axially Jahn-Teller-distorted Cu-O distance at 224.7 pm, a distinct Cu2Zn2superoxide dismutase mimetic activity was measured. The Cu-complex survived 600 μM bovine serum albumin and the thermodynamic stability (pK=17.4) was not very different from that of Cu-EDTA. The electronic absorption properties, circular dichroism and electron paramagnetism were in accordance with those of the type-II copper species.
Similar content being viewed by others
References
Bielski BHJ, Cabelli DE (1986) The role of transition metals in oxy-radical induced oxidation processes in biological systems. In: Rotilio G (ed) Superoxide and superoxide dismutase in chemistry, biology and medicine. Elsevier, Amsterdam, pp 3–8
Deuschle U, Weser U (1985) Copper and inflammation. Progr Clin Biochem Med 2:97–130
Fenton HJ, Jackson H (1899) The oxidation of polyhydric alcohols in presence of iron. J Chem Soc 75:1–11
Gärtner A, Hartmann H-J, Weser U (1984) A simple, rapid and efficient isolation of erythrocyte Cu2Zn2superoxide dismutase. Biochem J 221:549–551
Gärtner A, Weser U (1986) Molecular and functional aspects of superoxide dismutases. Top Curr Chem 132:1–61
Haber F, Weiss J (1934) The catalytical decomposition of hydrogen peroxide by iron salts. Proc R Soc Lond Ser A 147:332–351
Hartmann H-J, Gärtner A, Weser U (1985) Copper dependent control of the enzymic and phagocyte induced degradation of some biopolymers, a possible link to systemic inflammation. Clin Chim Acta 152:95–103
Holde KE van (1967) Physical studies of hemocyanins. III. Circular dichroism and absorption spectra. Biochemistry 6:93–99
Irving H, Mellor DH (1955) A new application of the theory of corresponding solutions to measurements of the stability of some metal complexes. J Chem Soc 131:3457–3462
Linss M, Weser U (1986) The di-Schiff-base by pyridine-2aldehyde and 1,4-diaminobutane, a flexible Cu(I)/Cu(II) chelator of significant superoxide dismutase mimetic activity. Inorg Chim Acta 125:117–121
Linss M, Weser U (1987) Redox behaviour and stability of active centre analogues of Cu2Zn2superoxide dismutase. Inorg Chim Acta 138:175–178
Naegeli HU, Zaehner H (1980) Stoffwechselprodukte von Mikroorganismen. Ferrithiocin. Helv Chim Acta 63:1400–1408
Peter HH (1985) Industrial aspects of iron chelators: pharmaceutical applications. In: Spik G, Montreuil J, Crichton RR, Mazurier J (eds) Proteins of iron storage and transport. Elsevier, Amsterdam, pp 293–303
Reinhammar B, Malmström BG (1981) ‘Blue’ copper-containing oxidases. In: Spiro TG (ed) Copper proteins, metal ions in biology, vol 3. Wiley, New York, pp 109–149
Younes M, Weser U (1976) Reactivity of superoxide dismutase-active Cu(II) complexes on the rate of adrenochrome formation. FEBS Lett 61:209–212
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schechinger, T., Hiller, W., Maichle, C. et al. A five-coordinate copper complex with superoxide dismutase mimetic activity fromStreptomyces antibioticus . Biol Metals 1, 112–116 (1988). https://doi.org/10.1007/BF01138069
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01138069