Abstract
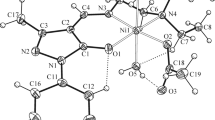
A 1∶1-complex of trinitrophenol with benzyldimethylamine (1) and a 1∶1∶1-complex, the product of the interaction of benzyldimethylamine, glycidyl phenyl ether, and phenol (2), have been synthesized and characterized by means of X-ray analysis. Complex1 is a precursor for the catalysts of selective isocyanate cyclotrimerization, while complex2 is one of those catalysts. In the crystal structure complex1 forms H-bonded cationic-anionic aggregates with proton transfer from phenol to the N-atom of the tertiary amine. Complex2 crystallizes as a monohydrate (2a) with a strong H-bond between the quaternary ammonium and phenolic components, and exists in crystals in the form of globular H-bonded dimeric hydrates. Possible pathways of dissociation of complex2a are discussed. The data obtained may be used as a basis for further interpretation of the regularities of cyclotrimerization of isocyanates catalyzed by complexes of this type.
Similar content being viewed by others
References
G.M.Pogosyan, V.A.Pankratov, V.N.Zaplishnyi, and S.G.Matsoyan,Polytriazines, Armenian Academy of Sciences, Erevan, 1987.
A.K.Zhitinkina, N.A.Shibanova, and O.G.Tarakanov,Uspekhi Khimii, 1985,54, 1866 [Rus.Chem.Rev., 1985 (Engl.Transl.)].
J.I.Jones, N.G.Savill,J.Chem.Soc., 1957, 4392.
W.E.Erner, US Patent 3010963.
B.D.Beitchman,Ind.Eng.Chem., 1966,58, 101.
S.V.Lindeman, Yu.T.Struchkov, L.P.Poddubnykh, and S.G.Dmitrienko,Koordinats. Khim., 1988, 14, 955 [Coordination Chemistry, 1988 (Engl.Transl.)].
S.V.Lindeman, Yu.T.Struchkov, B.F.Malichenko, V.E.Shklover, and V.A.Vasnev,Macromol. Chem., 1987,188, 1521.
Author information
Authors and Affiliations
Additional information
Translated fromIzvestiya Akademii Nauk. Seriya Khimicheskaya, No 1, pp. 92–97, January, 1993.
Rights and permissions
About this article
Cite this article
Pankratov, V.A., Frenkel, T.M., Shvorak, A.E. et al. The preparation and X-ray structural study of complexes that catalyze isocyanate cyclotrimerization. Russ Chem Bull 42, 81–87 (1993). https://doi.org/10.1007/BF00699980
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00699980