Abstract
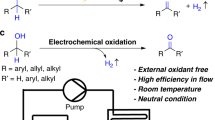
The key step in the electrochemical oxidation of ketoximes which determines the composition of the electrolysis products is the reaction of the intermediate oxoimmonium cation with the original oxime, whereas in the case of aldoximes, the key step is dimerization of the iminoxyl radicals.
Similar content being viewed by others
Literature cited
V. A. Petrosyan, M. E. Niyazymbetov, and é. V. Ul'yanova, Izv. Akad. Nauk SSSR, Ser. Khim., No. 7, 1683 (1989).
V. A. Petrosyan, M. E. Niyazymbetov, and é. V. Ul'yanova, Izv. Akad. Nauk SSSR, Ser. Khim., No. 7, 1687 (1989).
R. N. Butler, F. L. Scott, and T. A. F. O'Mahoney, Chem. Rev.,73, 93 (1973).
W. M. Lauer and W. S. Dyer, J. Am. Chem. Soc.,64, 1453 (1942).
W. H. Hunter and W. S. Dyer, J. Am. Chem. Soc.,55, 5053 (1933).
K. Anwers and H. Wunderling, Ber. Chem. Ges.,66, 538 (1933).
J. L. Brokenshire, J. R. Roberts, and K. U. Ingold, J. Am. Chem. Soc.,94, 7040 (1972).
é. G. Rozantsev and V. D. Sholle, The Organic Chemistry of Free Radicals, [in Russian], Khimiya, Moscow (1979), pp. 228–231.
G. Just and K. Dahl, Tetrahedron,24, 5251 (1968).
F. Fichter and H. Ris, Helv. Chim. Acta,7, 803 (1924).
H. C. Araujo, G. A. L. Ferreira, and J. R. Mahajan, J. Chem. Soc., Perkin Trans, 1, 2257 (1974).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 3, pp. 625–629, March, 1990.
Rights and permissions
About this article
Cite this article
Petrosyan, V.A., Niyazymbetov, M.E. & Ul'yanova, é.V. Anodic electrolysis of ketoximes and aldoximes: Mechanism. Russ Chem Bull 39, 546–550 (1990). https://doi.org/10.1007/BF00959580
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00959580