Abstract
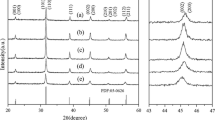
Shaped tetragonal barium titanate (BaTiO3) particles were prepared by the sol-emulsion-gel method with systematic variation of the concentration of a non-ionic surfactant in the organic phase (“oil phase”) of water-in-oil (w/o) emulsions above or below the critical micelle concentration (CMC). An acetate-based sol with equivalent oxide ratio BaO : TiO2 = 1 : 1 was selected for the study. Micelle formation of the surfactant and their self-aggregation apparently played an important role in controlling the shape and size of the droplets and accordingly shape and size of the final barium titanate particles resulting therefrom. Transmission electron microscopy (TEM) revealed that with low concentration of the surfactant (2–5 vol%) ill-formed spheres were obtained. Spherical particles were formed with increasing the surfactant concentration to 7 vol%. At 12 vol% of the surfactant, rod-like particles were first found to form, which with further increase in surfactant concentration to 15–20 vol%, gradually transformed to cube-like/lamellar type particles. Attempts have been made to explain the various particle shapes in terms of surfactant aggregation.
Similar content being viewed by others
References
M. Chatterjee, B. Siladitya, and D. Ganguli, Mater. Lett. 25, 261 (1995).
C.-H. Lu and C.-H. Yeh, Mater. Lett. 33, 129 (1997).
B. Siladitya, M. Chatterjee, and D. Ganguli, J. Sol-Gel Sci. Tech. 15, 271 (1999).
C.-H. Lu and Y.-P. Wu, Mater. Lett. 27, 13 (1996).
S.D. Ramamurthi, Z. Xu, and D.A. Payne, J. Amer. Ceram. Soc. 73, 2760 (1990).
J. Ray, M. Chatterjee, and D. Ganguli, J. Mater. Sci. Lett. 12, 1755 (1993).
T. Masui, K. Fujiwara, K. Machida, and G. Adachi, Chem. Mater. 9, 2197 (1997).
M. Chatterjee, J. Ray, A. Chatterjee, D. Ganguli, S.V. Joshi, and M.P. Srivastava, J. Mater. Sci. 28, 2803 (1993).
M. Chatterjee, D. Enkhtuvshin, B. Siladitya, and D. Ganguli, J. Mater. Sci. 33, 4937 (1998).
E. Ponthieu, E. Payen, and J. Grimblot, J. Non-Cryst. Solids 147 & 148, 598 (1992).
M. Bourrel and R.S. Schechter, Microemulsions and Related Systems (Marcel Dekker, New York, 1988).
E. Dickinson, in Controlled Particle,Droplet and Bubble Formation, edited by D.J. Wedlock (Butterworth-Heinemann, Oxford, 1994), p. 204.
D. Ganguli and M. Chatterjee, Ceramic Powder Preparation: A Handbook (Kluwer Academic Publishers, Boston, 1997).
J.D.F. Ramsay, in Sol-Gel Processing of Advanced Ceramics, edited by F.D. Gnanam (Oxford & IBH publishing Co. Pvt. Ltd., New Delhi, 1996), p. 47.
K. Borisch, S. Diele, P. Goring, H. Kresse, and C. Tschierske, J. Mater. Chem. 8, 529 (1998).
P. Behrens, A. Glaue, C. Haggenmuller, and G. Schechner, Solid State Ionics 101–103, 255 (1997).
H. Gutmann and A.S. Kertes, J. Colloid Interface Sci. 51, 406 (1975).
E. Ruckenstein and R. Nagarajan, J. Phys. Chem. 84, 1349 (1980).
G. Lindblom and L. Rilfors, Biochim. Biophys. Acta 988, 221 (1989).
C.A. Veracini and D. Catalano, in The Molecular Dynamics of Liquid Crystals, edited by G.R. Luckhurst and C.A. Veracini (Kluwer Academic Publishers, Netherlands, 1994) p. 505.
P.D. Godbole, S.B. Deshpande, H.S. Potdar, and S.K. Date, Mater. Lett. 12, 97 (1991).
J.M. Phelps and O. Ratsep, Ceram. Eng. Sci. Proc. 5, 1004 (1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chatterjee, M., Naskar, M. & Ganguli, D. Sol-Emulsion-Gel Synthesis of Shaped Powders in the System BaO-TiO2. Journal of Sol-Gel Science and Technology 16, 143–149 (1999). https://doi.org/10.1023/A:1008777524591
Issue Date:
DOI: https://doi.org/10.1023/A:1008777524591