Abstract
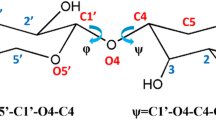
The conformational space of methyl 6-O-[(R)- and (S)- 1-carboxyethyl]-α-D-galactopyranoside has been investigated. A grid search employing energy minimization at each grid point over the three major degrees of freedom, namely φ, ψ and ω, identified low energy regions. The R-isomer shows five low energy conformers within ca. 1 kcal mol−1 of the global energy minimum. The S-isomer has two conformers within a few tenths of a kcal mol−1 of the global energy minimum. Langevin dynamics simulations have been have been performed at 300 K for 30 ns of each isomer. The φ dihedral angle has as its major conformer (g−1) for the R-isomer whereas it is the (g+) conformer for the S-isomer. For the ψ dihedral angle the (t) conformer has the highest population for both isomers. The dihedral angle ω has the (g+) conformer most highly populated, both for the R- and S-isomer. The above five and two conformational states for the R- and S-isomers, respectively, make up 90% in each case of the populated states during the Langevin dynamics (LD) simulations. Rate constants for the ω dihedral angle have been calculated based on a number correlation function. Three bond homo- and heteronuclear, i.e. proton and carbon-13, coupling constants have been calculated from the dynamics trajectories for comparison to experimental values. The heteronuclear coupling constant H2′,C6 has been measured for the S-isomer and found to be 3.3 Hz. The J value calculated from the LD simulations, namely 2.6 Hz, is in fair agreement with experiment. A comparison to the X-ray structure of the R-isomer shows that the conformation of the crystalline compound occupies the low energy region most highly populated as a single R-conformer (30%) during the LD simulations.
Similar content being viewed by others
References
Andersson M, Ratnayake S, Kenne L, Eriksson L, Stack RJ (1993) Carbohydrate Res 246: 291.
Andersson M, Kenne L, Stenutz R, Widmalm G (1994) Carbohydrate Res 254: 35.
Eriksson L, Pilotti Å, Stenutz R, Widmalm G (1996) Acta Cryst Sect C52: 2285.
Gasteiger J, Marsili M (1980) Ttrahedron 36: 3219.
Venable RM, Pastor RW (1988) Biopolymers 27: 1001
Nishida T, Widmalm G, Sandor P, (1996) Magn Reson Chem 34: 377.
Nishida T, Widmalm G, Sandor P (1995) Magn Reson Chem 33: 596.
McIntyre L, Freeman R (1992) J Magn Reson 96: 425.
del Rio-Portilla F, Blechta V, Freeman R (1994) J Magn Reson Ser A 111: 132.
Haasnoot CAG, de Leeuw FAAM, Altona C (1980) Tetrahedron 36: 2783.
Tvaroska I, Hricovíni M, Petráková E (1989) Carbohydrate Res 189: 359.
Widmalm G, Pastor RW (1992) J Chem Soc Faraday Trans 88: 1747.
Hardy BJ, Egan W, Widmalm G(1995) Int J Biol Macromol 17: 149.
Loncharich RJ, Brooks BR, Pastor RW (1992) Biopolymers 32: 523.
Pastor RW, Karplus M (1989) J Chem Phys 91: 211.
Zhou H-X (1989) Chem Phys Lett 164: 285.
Zwanzig R, Ailawadi A (1969) Phys Rev 182: 280.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stenutz, R., Widmalm, G. Conformational analysis of methyl 6-O-[(R)- and (S)-1-carboxyethyl]-α-D-galactopyranoside by MM and Langevin dynamics simulations. Glycoconj J 14, 973–981 (1997). https://doi.org/10.1023/A:1018527129939
Issue Date:
DOI: https://doi.org/10.1023/A:1018527129939