Abstract
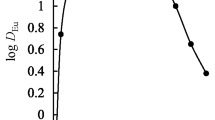
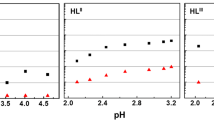
The co-ordination of plutonyl ions with 2-thenoyltrifluoroacetone (HTTA) has been investigated by distribution studies of239Pu between benzene and aqueous perchlorate medium at 25°C in order to understand the nature of the extracted species and their stability. The formation constants of the first and second complexes have been evaluated by Rydberg's graphical method as well as by least squares analysis of the distribution data using a computer programme. The overall formation constant (β2) for Pu(VI)-HTTA system has been found to be of the order of 1012.
Similar content being viewed by others
References
H. IRVING, D. N. EDGINGTON, J. Inorg. Nucl. Chem., 15 (1960) 158, 20 (1961) 314.
J. P. SHUKLA, V. K. MANCHANDA, M. S. SUBRAMANIAN, J. Electroanal. Chem., 50 (1974) 253.
J. J. KATZ, G. T. SEABORG, The Chemistry of the Actinide Elements, Methuen, 1957. p. 328.
J. RYDBERG, Arkiv. Kemi., 8 (1955) 101.
J. RYDBERG, Arkiv. Kemi., 8 (1955) 113.
J. RYDBERG, Acta Chem. Scand., 4 (1950) 1503.
D. DYRSSEN, Svensk. Kem. Tidskr., 68 (1956) 212.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shukla, J.P., Pai, S.A. & Subramanian, M.S. Investigation of the complex formation of plutonium(VI) with 2-thenoyltrifluoroacetone by solvent extraction. J. Radioanal. Chem. 34, 241–248 (1976). https://doi.org/10.1007/BF02519572
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02519572