Abstract
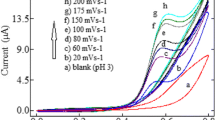
A novel voltammetric method for the determination of trifluralin is proposed based on a composit of carbon paste and copper nanowire as a sensitive sensor. The presence of copper nanowire in the composite film enhance the conductivity and as a result increased the electron transfer rate constant and so the current will increase. The composite exhibits a promising higher electrocatalytic activity towards the oxidation of trifluralin in pH 4.0 aqueous solution. The reduction peak currents of trifluralin increased remarkably and the reduction peak potential shifted positively at the nanostructuring electrode, compared with that at a bare CPE which show the enhanced effect of nanowire. The support electrolyte to provide a more defined and intense peak current for trifluraline determination was 0.05 mol L−1 phosphate buffer at pH 4.0. The fast Fourier transform square wave voltammetry was used as a new electrochemical technique in flow injection system to abtain more sensitivity by application of discrete fast Fourier transform method by background subtraction and two-dimensional integration of the electrode response over a selected potential range and time window, the signal-to-noise ratio has significantly increased and made the sensivity to be higher than other methods. The effective parameters such as frequency, amplitude, and pH were optimized to get the best sensitivity. As a result, the sensor showed a valuable response in linear concentration range of 100–0.02 nmol L−1 with a (limit of detection) LOD of 0.008 nmol L−1 and (limit of quantification) LOQ of 0.15 nmol L−1 for trifluralin. A good recovery was obtained for assay spiked urine samples and a good quantification of trifluralin was achieved in soil samples.








Similar content being viewed by others
References
Ashton FM, Crafts AS (1981) Dinitroanilines. In: Ashton FM, Crafts AS (eds) Mode of action of herbicides. Wiley, New York
Scheyer A, Morville S, Mirabel P, Millet M (2007) Atmospheric Envir 41:3604
Chernyak SM, Rice CP, Mc Connell LL (1996) Mar Pollut Bull 32:410–419
Hawthorne SB, Miller DJ, Louie PKK, Butler RD, Mayer GG (1996) J Environ Qual 25:594–600
Xiao YH, Li CM (2008) Electroanalysis 20:648–662
Luo XL, Morrin A, Killard AJ, Smyth MR (2006) Electroanalysis 18:319–326
Lu YH, Yang MH, Qu FL, Shen GL, Yu RQ (2007) Bioelectrochemistry 71:211–216
Shirsat MD, Too CO, Wallace GG (2008) Electroanalysis 20:150–156
Li DP, Frey MW, Baeumner AJ (2006) J Membr Sci 279:354–363
Yana S, Hea N, Songa Y, Zhanga Z, Qiana J, Xiao Z (2010) J Electroanal Chem 641:136–140
Zhang GJ, Zhang L, Huang MJ, Henry Luoa ZH, Ignatius TGK, Andy Lima EJ, Kanga TG, Chena Y (2010) Sens Actuators B 146:138–144
Duc HN, Van Quy N, Junga H, Kim D, Kima H, Honga SK (2010) Sens Actuators B 146:266–272
Na X, Yonglan D, Huihui A, Junjie F (2010) Microchim Acta 170:165–170
Wen X, Fei J, Chen X, Yi L, Ge F, Huang M (2008) Environ Pollut 156:1015–1020
Kalcher K, Kaufmann JM, Wang J, Svancara I, Vytras K, Neuhold C, Yang Z (1995) Electroanalysis 7:5–22
Valentini F, Amine A, Orlandocci S, Terranova ML, Palleschi G (2003) Anal Chem 75:5413–5421
Antiochia A, Lavagnini I, Magno F, Valentini F, Palleschi G (2004) Electroanalysis 16:1451–1458
Norouzi P, Ganjali MR, Shirvani-Arani S, Mohammadi A (2007) J Pharm Sci 95:893–904
Norouzi P, Shirvani-Arani S, Daneshgar P, Ganjali MR (2006) Biosens Bioelectron 22:1068–1074
Norouzi P, Ganjali MR, Moosavi-Movahedi AA (2007) J Braz Chem Soc 18:231–268
Norouzi P, Ganjali MR, Daneshgar P (2007) Anal Lett 40:547–559
Norouzi P, Ganjali MR, Hajiaghababaei L (2006) Anal Lett 39:1941–1953
Norouzi P, Ganjali MR, Daneshgar P (2007) Sens Actuators B 123:1125–1132
Norouzi P, Ganjali MR, Daneshgar P, Alizadeh T, Mohammadi A (2007) Anal Biochem 360:175–181
Norouzi P, Ganjali MR, Daneshgar P (2007) J Pharmacol Toxicol Methods 55:289–296
Norouzi P, Ganjali MR, Moosavi-movahedi AA, Larijani B (2007) Talanta 73:54–61
Norouzi P, Ganjali MR, Zare M, Mohammadi A (2007) J Pharm Sci 96:2009–2021
Norouzi P, Ganjali MR, Akbari-Adergani B (2007) Acta Chim Slov 53:499–505
Nabi Bidhendi G, Norouzi P, Daneshgar P, Ganjali MR (2007) J Hazard Mater 143:264–270
Norouzi P, Ganjali MR, Labbafi S, Mohammadi A (2007) Anal Lett 40:747–762
Norouzi P, Ganjali MR, Daneshgar P, Dinarvand P, Moosavi-Movahedi AA, Saboury AA (2007) Anal Chim Acta 590:74–80
Norouzi P, Nabi Bidhendi GR, Ganjali MR, Sepehri A, Ghorbani M (2005) Microchim Acta 152:123–129
Norouzi P, Ganjali MR, Alizadeh T, Daneshgar P (2006) Electroanalysis 18:947–954
Ganjali MR, Norouzi P, Ghorbani M, Sepehri A (2005) Talanta 66:1225–1233
Wang W, Lan Ch, Li Y, Hong K, Wang G (2002) Phys Lett 366:220–223
Dumanovic D, Ciric J (1973) Talanta 20:525–531
Kotoucek M, Opravilova M (1996) Anal Chim Acta 329:73–81
Zuman P, Fijalek Z, Dumanovic D, Suznjevic D (1992) Electroanalysis 4:783–794
Heyden YV, Nijhuis A, Smeyers-Verbeke J, Vandeginste BGM, Massaret DL (2001) J Pharm Biomed Anal 24:723–753
Miller JC, Miller JN (1984) Statistics for analytical chemistry. Ellis Horwood, Chichester 22:82–86
Bruzzoniti MC, Sarzanini C, Costantino G, Fungi M (2006) Anal Chim Acta 578:241
Vela N, Perez G, Navarro G, Navarro S (2007) J AOAC Int 90:544–549
Karasali H, Balayannis G, Hourdakis A, Ambrus A (2006) J Chromatogr A 1129:300–303
Acknowledgment
This work was supported by Research Council of University of Tehran, the International Foundation of Science (INS), and Iran National Science Foundation (INSF).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mirabi-semnakolaii, A., Daneshgar, P., Moosavi-Movahedi, A.A. et al. Sensitive determination of herbicide trifluralin on the surface of copper nanowire electrochemical sensor. J Solid State Electrochem 15, 1953–1961 (2011). https://doi.org/10.1007/s10008-010-1212-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-010-1212-8