Abstract
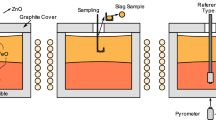
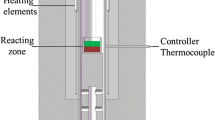
A kinetic model for the oxidation of selenium and tellurium in the molten bath of an industrial Kaldo furnace during the refining step is presented in this article. During plant experiments, samples of the molten metal were collected hourly and chemically analyzed for selenium, tellurium, and silver. Selenium oxidized from the beginning of the refining step, whereas oxidation of tellurium only occurred until the Se/Te mass ratio decreased to a value of 0.55. Good agreement between the model predictions and the experimental data was obtained.
Similar content being viewed by others
References
Precious Metals Plant Induction Manual (Nacozari, Mexico: Mexicana de Cobre, S.A. Internal Report, 1998) (in Spanish).
P.M. Devia and G.A. Luraschi, “Thermodynamics of the Ag-Se-O System at 1100°C,” Advances in Extractive Metallurgy 1997, ed. M. Certucha, L. Almazan, and J. Valenzuela (Hermosillo, Mexico: Universidad de Sonora, 1997), pp. 221–234 (in Spanish).
P.M. Devia, “Thermodynamics of the Ag-Te-O System at 1100°C,” Advances in Extractive Metallurgy 2003, ed. S. Aguayo, M. Encinas, and A. Valenzuela (Hermosillo, Mexico: Universidad de Sonora, 2003) (CD in Spanish).
Microsoft® Excel (Redmond, WA: Microsoft Corporation, 2002).
Author information
Authors and Affiliations
Additional information
For more information, contact Manuel Perez-Tello, Universidad de Sonora, Departamento de Ingeniería Química y Metalurgia, Hermosillo, Sonora, Mexico 83000; +52-662-259-2107; fax +52-662-259-2107; e-mail mperezt@iq.uson.mx.
Rights and permissions
About this article
Cite this article
Perez-Tello, M., Sanchez-Corrales, V.M., Prieto-Sanchez, M.R. et al. A kinetic model for the oxidation of selenium and tellurium in an industrial kaldo furnace. JOM 56, 52–54 (2004). https://doi.org/10.1007/s11837-004-0236-x
Issue Date:
DOI: https://doi.org/10.1007/s11837-004-0236-x