Abstract
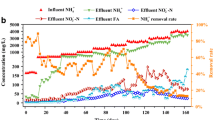
Performance and microbial community composition were evaluated in a two-phase anaerobic and aerobic system treating sulfate-rich cellulosic ethanol wastewater (CEW). The system was operated at five different chemical oxygen demand (COD)/SO4 2− ratios (63.8, 26.3, 17.8, 13.7, and 10.7). Stable performance was obtained for total COD removal efficiency (94.5%), sulfate removal (89.3%), and methane production rate (11.5 L/day) at an organic loading rate of 32.4 kg COD/(m3·day). The acidogenic reactor made a positive contribution to net VFAs production (2318.1 mg/L) and sulfate removal (60.9%). Acidogenic bacteria (Megasphaera, Parabacteroides, unclassified Ruminococcaceae spp., and Prevotella) and sulfate-reducing bacteria (Butyrivibrio, Megasphaera) were rich in the acidogenic reactor. In the methanogenic reactor, high diversity of microorganisms corresponded with a COD removal contribution of 83.2%. Moreover, methanogens (Methanosaeta) were predominant, suggesting that these organisms played an important role in the acetotrophic methanogenesis pathway. The dominant aerobic bacteria (Truepera) appeared to have been responsible for the COD removal of the SBR. These results indicate that dividing the sulfate reduction process could effectively minimize sulfide toxicity, which is important for the successful operation of system treating sulfate-rich CEW.



Similar content being viewed by others
References
Ahring BK, Ibrahim AA, Mladenovska Z (2001) Effect of temperature increase from 55 to 65 C on performance and microbial population dynamics of an anaerobic reactor treating cattle manure. Water Res 35:2446–2452
APHA, AWWA, WEF (2005) Standard methods for the examination of water and wastewater. American Public Health Association, Washington
Berner RA (1963) Electrode studies of hydrogen sulfide in marine sediments. Geochim Cosmochim A 27:563–575
Briones AM, Daugherty BJ, Angenent LT, Rausch K, Tumbleson M, Raskin L (2009) Characterization of microbial trophic structures of two anaerobic bioreactors processing sulfate-rich waste streams. Water Res 43:4451–4460
Choeisai P, Jitkam N, Silapanoraset K, Yubolsai C, Yoochatchaval W, Yamaguchi T, Onodera T, Syutsubo K (2014) Sugarcane molasses-based bio-ethanol wastewater treatment by two-phase multi-staged up-flow anaerobic sludge blanket (UASB) combination with up-flow UASB and down-flow hanging sponge. Water Sci Technol 69:1174–1180
Demirel B, Scherer P (2008) The roles of acetotrophic and hydrogenotrophic methanogens during anaerobic conversion of biomass to methane: a review. Rev Environ Sci Bio 7:173–190
Demirel B, Yenigün O (2002) Two-phase anaerobic digestion processes: a review. J Chem Technol Biot 77:743–755
Eichorst SA, Joshua C, Sathitsuksanoh N, Singh S, Simmons BA, Singer SW (2014) Substrate-specific development of thermophilic bacterial consortia by using chemically pretreated switchgrass. Appl Environ Microbiol 80:7423–7432
Emery R, Smith C, To LF (1957) Utilization of inorganic sulfate by rumen microorganisms: II. The ability of single strains of rumen bacteria to utilize inorganic sulfate 1, 2. Appl Microbiol 5:363–366
Fox P, Pohland FG (1994) Anaerobic treatment applications and fundamentals—substrate-specificity during phase-separation. Water Environ Res 66:716–724
Göker M, Saunders E, Lapidus A, Nolan M, Lucas S, Hammon N, Deshpande S, Cheng J-F, Han C, Tapia R (2012) Genome sequence of the moderately thermophilic, amino-acid-degrading and sulfur-reducing bacterium Thermovirga lienii type strain (Cas60314 T). Stand Genomic Sci 6:230
Greene AC, Patel BK, Yacob S (2009) Geoalkalibacter subterraneus sp. nov., an anaerobic Fe(III)- and Mn(IV)-reducing bacterium from a petroleum reservoir, and emended descriptions of the family Desulfuromonadaceae and the genus Geoalkalibacter. Int J Syst Evol Microbiol 59:781–785
Guo Z, Zhou A, Yang C, Liang B, Sangeetha T, He Z, Wang L, Cai W, Wang A, Liu W (2015) Enhanced short chain fatty acids production from waste activated sludge conditioning with typical agricultural residues: carbon source composition regulates community functions. Biotechnol Biofuels 8:1
Hino T, Miyazaki K, Kuroda S (1991) Role of extracellular acetate in the fermentation of glucose by a ruminal bacterium, Megasphaera elsdenii. J Gen Appl Microbiol 37:121–129
Huws SA, Kim EJ, Lee MR, Scott MB, Tweed JK, Pinloche E, Wallace RJ, Scollan ND (2011) As yet uncultured bacteria phylogenetically classified as Prevotella, Lachnospiraceae incertae sedis and unclassified Bacteroidales, Clostridiales and Ruminococcaceae may play a predominant role in ruminal biohydrogenation. Environ Microbiol 13:1500–1512
Isa Z, Grusenmeyer S, Verstraete W (1986) Sulfate reduction relative to methane production in high-rate anaerobic digestion—microbiological aspects. Appl Environ Microbiol 51:580–587
Jimenez J, Gonidec E, Rivero JAC, Latrille E, Vedrenne F, Steyer J-P, Bakhrouf A (2014) Prediction of anaerobic biodegradability and bioaccessibility of municipal sludge by coupling sequential extractions with fluorescence spectroscopy: towards ADM1 variables characterization. Water Res 50:359–372
Kalyuzhnyi S, Fedorovich V, Lens P, Pol LH, Lettinga G (1998) Mathematical modelling as a tool to study population dynamics between sulfate reducing and methanogenic bacteria. Biodegradation 9:187–199
Kim SB, Lee Y (2002) Diffusion of sulfuric acid within lignocellulosic biomass particles and its impact on dilute-acid pretreatment. Bioresour Technol 83:165–171
Kühl M, Jørgensen BB (1992) Microsensor measurements of sulfate reduction and sulfide oxidation in compact microbial communities of aerobic biofilms. Appl Environ Microbiol 58:1164–1174
Lee GH, Rhee MS, Chang DH, Lee J, Kim S, Yoon MH, Kim BC (2013) Oscillibacter ruminantium sp. nov., isolated from the rumen of Korean native cattle. Int J Syst Evol Microbiol 63:1942–1946
Lens P, Kuenen J (2001) The biological sulfur cycle: novel opportunities for environmental biotechnology. Water Sci Technol 44:57–66
Lens PNL, Visser A, Janssen AJH, Pol LWH, Lettinga G (1998) Biotechnological treatment of sulfate-rich wastewaters. Crit Rev Env Sci Tech 28:41–88
Lykidis A, Chen C-L, Tringe SG, McHardy AC, Copeland A, Kyrpides NC, Hugenholtz P, Macarie H, Olmos A, Monroy O (2011) Multiple syntrophic interactions in a terephthalate-degrading methanogenic consortium. The ISME Journal 5:122–130
Montpart N, Rago L, Baeza JA, Guisasola A (2015) Hydrogen production in single chamber microbial electrolysis cells with different complex substrates. Water Res 68:601–615
Percheron G, Bernet N, Moletta R (1997) Start-up of anaerobic digestion of sulfate wastewater. Bioresour Technol 61:21–27
Pholchan MK, Baptista Jde C, Davenport RJ, Curtis TP (2010) Systematic study of the effect of operating variables on reactor performance and microbial diversity in laboratory-scale activated sludge reactors. Water Res 44:1341–1352
Reis M, Goncalves L, Carrondo M (1988) Sulphate removal in acidogenic phase anaerobic digestion. Environ Technol Lett 9:775–784
Sánchez-Andrea I, Stams AJ, Amils R, Sanz JL (2013) Enrichment and isolation of acidophilic sulfate-reducing bacteria from Tinto River sediments. Env Microbiol Rep 5:672–678
Shan L, Yu Y, Zhu Z, Zhao W, Wang H, Ambuchi JJ, Feng Y (2015) Microbial community analysis in a combined anaerobic and aerobic digestion system for treatment of cellulosic ethanol production wastewater. Environ Sci Pollut R 22:17789–17798
Shen PH, Zhang JY, Zhang J, Jiang CJ, Tang XL, Li JF, Zhang M, Wu B (2013) Changes in microbial community structure in two anaerobic systems to treat bagasse spraying wastewater with and without addition of molasses alcohol wastewater. Bioresour Technol 131:333–340
Shin H-S, Bae B-U, Lee J-J, Paik B-C (1992) Anaerobic digestion of distillery wastewater in a two-phase UASB system. War Sci Tech 25:361–371
Stams A, Plugge C, De Bok F, Van Houten B, Lens P, Dijkman H, Weijma J (2005) Metabolic interactions in methanogenic and sulfate-reducing bioreactors. Water Sci Technol 52:13–20
Tan H-Q, Li T-T, Zhu C, Zhang X-Q, Wu M, Zhu X-F (2012) Parabacteroides chartae sp. nov., an obligately anaerobic species from wastewater of a paper mill. Int J Syst Evol Microb 62:2613–2617
Vohra M, Manwar J, Manmode R, Padgilwar S, Patil S (2014) Bioethanol production: feedstock and current technologies. Journal of Environmental Chemical Engineering 2:573–584
Wang H, Hu C, Hu X, Yang M, Qu J (2012) Effects of disinfectant and biofilm on the corrosion of cast iron pipes in a reclaimed water distribution system. Water Res 46:1070–1078
Wilkie AC, Riedesel KJ, Owens JM (2000) Stillage characterization and anaerobic treatment of ethanol stillage from conventional and cellulosic feedstocks. Biomass Bioenergy 19:63–102
Zheng Y, Xiao Y, Yang Z-H, Wu S, Xu H-J, Liang F-Y, Zhao F (2014) The bacterial communities of bioelectrochemical systems associated with the sulfate removal under different pHs. Process Biochem 49:1345–1351
Acknowledgements
This research was supported by the National Key Research and Development Program of China (2016YFC0401101) and the National Natural Science Foundation of China (51308150, 41405130). The research also got the support from the National Science Technology Pillar Program, China (2015BAD15B0502) and the Heilongjiang Postdoctoral Fund (LBH-Z12132) in China. The authors also acknowledge the support from the State Key Laboratory of Urban Water Resource and Environment (2015DX08), and support from the Fundamental Research Funds for the Central Universities (grant no. HIT.MKSTISP.2016 14).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible editor: Diane Purchase
Electronic supplementary material
ESM 1
(DOCX 315 kb)
Rights and permissions
About this article
Cite this article
Shan, L., Zhang, Z., Yu, Y. et al. Performance of CSTR–EGSB–SBR system for treating sulfate-rich cellulosic ethanol wastewater and microbial community analysis. Environ Sci Pollut Res 24, 14387–14395 (2017). https://doi.org/10.1007/s11356-017-9022-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-9022-5