Abstract
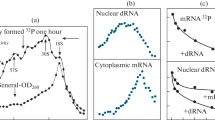
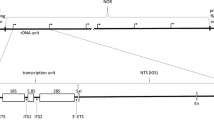
The DNA coding for 28 S and 18 S ribosomal RNA, including the spacer regions, has been isolated from calf (Bos taurus) thymus gland. The method used included shearing of the total DNA to a highly homogeneous size population, selective heat denaturation and S 1 nuclease treatment to remove single stranded DNA. Repeated centrifugation on density gradients yields a 140-fold purified rDNA fraction with a GC content of 61.2%. Eco RI nuclease cleaves this DNA into two fragments of 16.4 and 4.9×106 daltons. Hybridization of these fragments with 28 S and 18 S rRNA shows that the 28 S coding sequence is located mostly on the 4.9×106 dalton fragment, while both the 16.4 and 4.9×106 dalton fragments contain the 18 S sequence. The data indicate that the ribosomal RNA gene has a repeat unit of 21.3×106 daltons which includes a nontranscribed spacer of about 12.5×106 daltons.
Similar content being viewed by others
References
Birnstiel, M., Speirs, J., Purdom, I., Jones, K., Loaning, U.E.: Properties and composition of the isolated ribosomal DNA satellite of Xenopus laevis. Nature (Lond.) 219, 454–463 (1968)
Birnstiel, M., Telford, J., Weinberg, E., Stafford, D.: Isolation and some properties of the genes coding for histone proteins. Proc. nat. Acad. Sci. (Wash.) 71, 2900–2904 (1974)
Botchan, M.R.: Bovine satellite I DNA consists of repetitive units 1,400 base pairs in length. Nature (Lond.) 251, 288–292 (1974)
Bross, K., Krone, W.: On the number of ribosomal RNA genes in man. Humangenetik 14, 137–141 (1972)
Brown, D.D., Wensink, P.C., Jordan, E.: Purification and some characteristics of 5S DNA from Xenopus laevis. Proc. nat. Acad. Sci. (Wash.) 68, 3175–3179 (1971)
Chambon, P., Mandel, J.L., Gissinger, F., Kedinger, C., Gros-Bellard, M., Hossenloop, P.: Double-stranded viral and cellular DNA's as templates for purified mammalian DNA-dependent RNA polymerases. In: Regulation of transcription and translation in eukaryotes (E.K.F. Bautz, P. Karlson and H. Kersten, eds.), pp. 125–138. New York-Heidelberg-Berlin: Springer 1973
Dawid, I.B., Brown, D.D., Reeder, R.H.: Composition and structure of chromosomal and amplified ribosomal DNA's of Xenopus laevis. J. molec. Biol. 51, 341–360 (1970)
Gall, J.G.: Free ribosomal RNA genes in the macronucleus of Tetrahymena. Proc. nat. Acad. Sci. (Wash.) 71, 3078–3081 (1974)
Gall, J.G., Rochaix, J.-D.: The amplified ribosomal DNA of Dytiscid beetles. Proc. nat. Acad. Sci. (Wash.) 71, 1819–1823 (1974)
Greene, P.J., Betlach, M.C., Goodman, H.M., Boyer, H.W.: The EcoRI restriction endonuclease. In: Methods in molecular biology (R.B. Wickner, ed.), pp. 87–111. New York: Marcel Dekker Inc. 1974
Hamkalo, B.A., Miller, O.L., Jr.: Electron microscopy of genetic activity. Ann. Rev. Biochem. 42, 379–396 (1973)
Hastings, J.R.B., Kirby, K.S.: The nucleic acids of Drosophila melanogaster. Biochem. J. 100, 532–539 (1966)
Hayward, G.S.: Unique double-stranded fragments of bacteriophage T5 DNA resulting from preferential shear-induced breakage at nicks. Proc. nat. Acad. Sci. (Wash.) 71, 2108–2112 (1974)
Hearst, J.E., Vinograd, J.: The net hydration of T-4 bacteriophage deoxyribonucleic acid and the effect of hydration on buoyant behavior in a density gradient at equilibrium in the ultracentrifuge. Proc. nat. Acad. Sci. (Wash.) 47, 1005–1014 (1961)
Joseph, D.R., Stafford, D.W.: Purification of sea urchin ribosomal RNA genes with a single-strand specific nuclease. Biochim. biophys. Acta (Amst.) 418, 167–174 (1976)
Patterson, J.B., Stafford, D.W.: Sea urchin satellite deoxyribonucleic acid. Its large-scale isolation and hybridization with homologous ribosomal ribonucleic acid. Biochemistry (Wash.) 9, 1278–1283 (1970)
Reeder, R.H., Roeder, R.G.: Ribosomal RNA synthesis in isolated nuclei. J. molec. Biol. 67, 433–441 (1972)
Scherberg, N.H., Refetoff, S.: Hybridization of RNA labelled with 125I to high specific activity. Nature (Lond.) New Biology 242, 142–145 (1973)
Scherrer, K., Darnell, J.E.: Sedimentation characteristics of rapidly labelled RNA from HeLa cells. Biochem. biophys. Res. Commun. 7, 486–490 (1962)
Schildkraut, C., Lifson, S.: Dependence of the melting temperature of DNA in salt concentration. Biopolymers 3, 195–208 (1965)
Southern, E.M.: Detection of specific sequences among DNA fragments separated by gel electrophoresis. J. molec. Biol. 98, 503–517 (1975)
Suzuki, Y., Gage, L.P., Brown, D.D.: The genes for silk fibroin in Bombyx mori. J. molec. Biol. 70, 637–649 (1972)
Vogt, V.M.: Purification and further properties of single-strand-specific nuclease from Aspergillus oryzae. Europ. J. Biochem. 33, 192–200 (1973)
Wartell, R.M., Larson, J.E., Wells, R.D.: Netropsin. J. biol. Chem. 249, 6719–6731 (1974)
Weinberg, E.S., Overton, G.C., Shutt, R.H., Reeder, R.H.: Histone gene arrangement in the sea urchin, Strongylocentrotus purpuratus. Proc. nat. Acad. Sci. (Wash.) 72, 4815–4819 (1975)
Wellauer, P.K., Dawid, I.B., Kelley, D.W., Perry, R.: Secondary structure maps of ribosomal RNA. II. Processing of mouse L-cell ribosomal RNA and variations in the processing pathway. J. molec. Biol. 89, 397–407 (1974)
Wellauer, P.K., Reeder, R.H., Carroll, D., Brown, D.D., Deutsch, A., Higashinakagawa, T., Dawid, I.B.: Amplified ribosomal DNA from Xenopus laevis has heterogeneous spacer lengths. Proc. nat. Acad. Sci. (Wash.) 71, 2823–2827 (1974)
Wensink, P.C., Brown, D.D.: Denaturation map of the ribosomal DNA of Xenopus laevis. J. molec. Biol. 60, 235–247 (1971)
Wikman, J., Howard, E., Busch, H.: Studies on the primary structure of ribosomal 28S ribonucleic acid and its nucleolar precursors. J. biol. Chem. 244, 5471–5480 (1969)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Blin, N., Stephenson, E.C. & Stafford, D.W. Isolation and some properties of a mammalian ribosomal DNA. Chromosoma 58, 41–50 (1976). https://doi.org/10.1007/BF00293439
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00293439