Summary
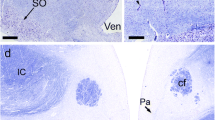
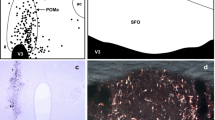
The GABAergic innervation of the mouse pituitary, including the median eminence, was studied at light microscopic and ultrastructural levels by use of a pre-embedding immunocytochemical technique with antibodies directed against GABA. In the median eminence, a high density of GABA-immunoreactive fibers was found in the external layer where the GABAergic varicosities were frequently observed surrounding the blood vessels of the primary capillary plexus. In the internal and subependymal layers, only few fibers were immunoreactive. The intense labeling of the external layer was observed in the entire rostro-caudal extent of the median eminence. In the pituitary proper, a dense network of GABA-immunoreactive fibers was revealed throughout the neural and intermediate lobes, entering via the hypophyseal stalk. The anterior and tuberal lobes were devoid of any immunoreactivity. The GABA-immunoreactive terminals were characterized in the median eminence, and in the intermediate and posterior lobes at the electron-microscopic level. They contained small clear vesicles, occasionally associated with dense-core vesicles or neurosecretory granules. In the intermediate lobe they were seen to be in contact with the glandular cells. In the posterior lobe and in the median eminence, GABA-immunoreactive terminals were frequently located in the vicinity of blood vessels. These results further support the concept of a role of GABA in the regulation of hypophyseal functions, via the portal blood for the anterior lobe, directly on the cells in the intermediate lobe, and via axo-axonic mechanisms in the median eminence and posterior lobe.
Similar content being viewed by others
References
Anderson RA, Mitchell R (1986a) Effects of γ-aminobutyric acid receptor agonists on the secretion of growth hormone, luteinizing hormone, adrenocorticotrophic hormone and thyroid-stimulating hormone from the rat pituitary gland in vitro. J Endocrinol 108:1–8
Anderson RA, Mitchell R (1986b) Distribution of GABA binding site subtypes in rat pituitary gland. Brain Res 365:78–84
Berod A, Chat M, Paul L, Tappaz M (1984) Catecholaminergic and GABAergic anatomical relationship in the rat substantia nigra, locus coeruleus, and hypothalamic median eminence: Immunocytochemical visualisation of biosynthetic enzymes on serial semithin plastic-embedded sections. J Histochem Cytochem 32:1331–1338
Bosler O, Calas A (1982) Radioautographic investigation of monoaminergic neurons: An evaluation. Brain Res Bull 9:151–169
Calas A (1985) Morphological correlates of chemically specified neuronal interactions in the hypothalamo-hypophyseal area. Neurochem Int 7:927–940
Demeneix BA, Desaulles E, Feltz P, Loeffler JP (1984) Dual population of GABAa and GABAb receptors in rat pars intermedia demonstrated by release of alpha MSH caused by barium ions. Br J Pharmacol 82:183–190
Fuxe K, Hökfelt T, Johannson O, Ganten D, Goldstein M, Perez de la Mora M, Possani L, Tapia R, Teran L, Palacios R, Said S, Mutt V (1976) Monoamine neuron systems in the hypothalamus and their relation to the GABA and peptide-containing neurons. In: Mornex R, Barry J (eds) Neuromediateurs ct polypeptides hypothalamiques. Une action relachante ou inhibitrice. Institut National et de la Recherche Médicale, Paris, pp 17–40
Gamrani H, Harandi M, Belin MF, Dubois MP, Calas A (1984) Direct electron microscopic evidence for the coexistence of GABA uptake and endogenous serotonin in the same rat central neurons by coupled radioautographic and immunocytochemical procedures. Neurosci Lett 48:25–30
Gamrani H, Onteniente B, Seguela P, Geffard M, Calas A (1986) Gamma-aminobutyric acid-immunoreactivity in the rat hippocampus. A light and electron microscopic study with anti-GABA antibodies. Brain Res 364:30–38
Grandison L, Guidotti A (1979) γ-Aminobutyric acid receptor function in rat anterior pituitary: evidence for control of prolactin release. Endocrinology 105:754–759
Gudelsky GA, Apud JA, Masotto C, Locatelli V, Cocchi D, Racagni G, Müller EE (1983) Ethanolamine-O-sulfate enhances γ-aminobutyric acid secretion into hypophyseal portal blood and lowers serum prolactin concentrations. Neuroendocrinology 37:397–399
Hamberger A, Norström A, Sandberg M, Svanberg U (1979) In vitro GABA transport in the neurohypophysis from rats with hereditary diabetes insipidus and after osmotic stimulation. Brain Res 174:341–344
Hendry SHC, Jones EG, DeFelipe J, Schmechel D, Brandon C, Emson PC (1984) Neuropeptide-containing neurons of the cerebral cortex are also GABAergic. Proc Natl Acad Sci USA 81:6526–6530
Jordan D, Poncet C, Veisseire M, Mornex R (1983) Role of GABA in the control of thyrotropin secretion in the rat. Brain Res 268:105–110
Locatelli V, Cocchi D, Frigerio C, Betti R, Krogsgaard-Larsen P, Racagni G, Müller EE (1979) Dual γ-aminobutyric acid control of prolactin secretion in the rat. Endocrinology 105:778–785
Mathison RD, Dreifuss JJ (1980) Structure-activity relationships of a neurohypophysial GABA receptor. Brain Res 187:476–480
McCann SM, Vijayan E, Negro-Vilar A, Mizunuma H, Mangat H (1984) Gamma-aminobutyric acid (GABA), a modulator of anterior pituitary hormone secretion by hypothalamic and pituitary action. Psychoneuroendocrinology 9:97–106
Mulchahey JJ, Neill JD (1982) Gamma aminobutyric acid (GABA) levels in hypophyseal stalk plasma of rats. Life Sci 31:453–456
Oertel WH, Mugnaini E, Tappaz ML, Weise VK, Dahl AL, Schmechel DE, Kopin IJ (1982) Central GABAergic innervation of neurointermediate pituitary lobe: Biochemical and immunocytochemical study in the rat. Proc Natl Acad Sci USA 79:675–679
Ondo JG (1974) Gamma-aminobutyric acid effects on pituitary gonadotropin-secretion. Science 186:738–739
Onteniente B, Geffard M, Campistron G, Calas A (1986) An ultrastructural study of GABA-immunoreactive neurons and terminals in the septum of the rat. J Neurosci (in press)
Pass KA, Ondo JG (1977) The effects of γ-aminobutyric acid on prolactin and gonadotropin secretion in the unanaesthetized rat. Endocrinology 100:1437–1442
Perez de La Mora M, Possani LD, Tapia R, Teran L, Palacios R, Fuxe K, Hökfelt T, Ljungdahl A (1981) Demonstration of central GABA nerve terminals by means of antibodies against glutamate decarboxylase. Neuroscience 6:875–895
Peters LL, Hoefer MT, Ben-Jonathan N (1981) The posterior pituitary: Regulation of anterior pituitary prolactin secretion. Science 213:659–661
Rabhi M, Onteniente B, Geffard M, Calas A (1985) Innervation GABAergique de la région hypothalamo-hypophysaire. Etude immunocytochimique chez la souris à l'aide d'anticorps anti-GABA. Ann Endocrinol 46:46 [Abstr]
Racagni G, Apud JA, Locatelli V, Cocchi D, Nistico' G, di Giorgio RM, Müller EE (1979) GABA of CNS origin in the rat anterior pituitary inhibits prolactin secretion. Nature 281:575–578
Racagni G, Apud JA, Cocchi D, Locatelli V, Müller EE (1982) Minireview GABAergic control of anterior pituitary hormone secretion. Life Sci 31:823–838
Racagni G, Apud JA, Cocchi D, Locatelli V, Iuliano E, Casanueva F, Müller EE (1984) Regulation of prolactin secretion during suckling: involvement of the hypothalamo-pituitary GABAergic system. J Endocrinol Invest 7:481–487
Rioch DME, Wislocki GB, O'Leary TL (1940) A precis of preoptic, hypothalamic and hypophyseal terminology with atlas. Proc Ass Res Nerv Ment Dis 20:1–30
Seguela P, Geffard M, Buijs RM, Le Moal M (1984) Antibodies against γ-aminobutyric acid: Specificity studies and immunocytochemical results. Proc Natl Acad Sci USA 81:3888–3892
Seguela P, Gamrani H, Geffard M, Calas A, Le Moal M (1985) Ultrastructural immunocytochemistry of γ-aminobutyrate in the cerebral and cerebellar cortex of the rat. Neuroscience 16:865–874
Stoeckel ME, Tappaz M, Hindelang C, Seweryn C, Porte A (1985) Opposite effects of monosodium glutamate on the dopaminergic and GABAergic innervations of the median eminence and the intermediate lobe in the mouse. Neurosci Lett 56:249–255
Tappaz ML (1984) GABA and anterior pituitary function: anatomical data. Psychoneuroendocrinology 9:85–94
Tappaz ML, Brownstein ML, Kopin IJ (1977) Glutamate decarboxylase (GAD) and γ-aminobutyric acid (GABA) in discrete nuclei of hypothalamus and substantia nigra. Brain Res 125:109–121
Tappaz M, Aguera M, Belin MC, Pujol JF (1980) Autoradiography of GABA in the hypothalamic median eminence. Brain Res 186:379–391
Tappaz ML, Oertel WH, Wassef M, Mugnaini E (1982) Central GABAergic neuroendocrine regulations: Pharmacological and morphological evidence. In: Buijs RM, Pévet P, Swaab K.F (eds) Chemical Transmission in the Brain, Progress in Brain Res Elsevier Biochemical Press 55:77–96
Tappaz ML, Wassef M, Oertel WH, Mugnaini E (1983a) GABA and adenohypophyseal function: Immunocytochemical evidence. In: E Endröczi (ed) Integrative Neurohumoral Mechanisms, Elsevier Science Publishers, pp 310–330
Tappaz ML, Wassef M, Oertel WH, Paut L, Pujol JF (1983b) Light and electron-microscopic immunocytochemistry of glutamic acid decarboxylase (GAD) in the basal hypothalamus: morphological evidence for neuroendocrine γ-aminobutyrate (GABA). Neuroscience 9:271–287
Taraskevich PS, Douglas WW (1982) GABA directly affects electrophysiological properties of pituitary pars intermedia cells. Nature 299:733–734
Tomiko SA, Taraskevich PS, Douglas WW (1983) GABA acts directly on cells of pituitary pars intermedia to alter hormone output. Nature 301:706–707
Van der Heyden JAM, De Kloet ER, Korf J, Versteeg DHG (1979) GABA content of discrete brain nuclei and spinal cord of the rat. J Neurochem 33:857–861
Vijayan E, McCann SM (1978) The effects of intraventricular injection of γ-aminobutyric acid (GABA) on prolactin and gonadotropin release in conscious female rats. Brain Res 155:35–43
Vincent SR, Hökfelt T, Wu JY (1982) GABA neuron systems in hypothalamus and the pituitary gland. Immunohistochemical demonstration using antibodies against glutamate decarboxylase. Neuroendocrinology 34:117–125
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rabhi, M., Onteniente, B., Kah, O. et al. Immunocytochemical study of the GABAergic innervation of the mouse pituitary by use of antibodies against gamma-aminobutyric acid (GABA). Cell Tissue Res. 247, 33–40 (1987). https://doi.org/10.1007/BF00216544
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00216544