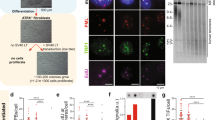
Abstract
Telomere dysfunction is typically studied under conditions in which a component of the six-subunit shelterin complex that protects chromosome ends is disrupted. The nature of spontaneous telomere dysfunction is less well understood. Here we report that immortalized human cell lines lacking wild-type p53 function spontaneously show many telomeres with a DNA damage response (DDR), commonly affecting only one sister chromatid and not associated with increased chromosome end-joining. DDR+ telomeres represent an intermediate configuration between the fully capped and uncapped (fusogenic) states. In telomerase activity–positive (TA+) cells, DDR is associated with low TA and short telomeres. In cells using the alternative lengthening of telomeres mechanism (ALT+), DDR is partly independent of telomere length, mostly affects leading strand–replicated telomeres, and can be partly suppressed by TRF2 overexpression. In ALT+ (but not TA+) cells, DDR+ telomeres preferentially associate with large foci of extrachromosomal telomeric DNA and recombination proteins. DDR+ telomeres therefore arise through different mechanisms in TA+ and ALT+ cells and have different consequences.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Palm, W. & de Lange, T. How shelterin protects mammalian telomeres. Annu. Rev. Genet. 42, 301–334 (2008).
Takai, H., Smogorzewska, A. & de Lange, T. DNA damage foci at dysfunctional telomeres. Curr. Biol. 13, 1549–1556 (2003).
Denchi, E.L. & de Lange, T. Protection of telomeres through independent control of ATM and ATR by TRF2 and POT1. Nature 448, 1068–1071 (2007).
Guo, X. et al. Dysfunctional telomeres activate an ATM-ATR-dependent DNA damage response to suppress tumorigenesis. EMBO J. 26, 4709–4719 (2007).
Smogorzewska, A. & de Lange, T. Different telomere damage signaling pathways in human and mouse cells. EMBO J. 21, 4338–4348 (2002).
Karlseder, J., Broccoli, D., Dai, Y., Hardy, S. & de Lange, T. p53- and ATM-dependent apoptosis induced by telomeres lacking TRF2. Science 283, 1321–1325 (1999).
d'Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003).
Smogorzewska, A., Karlseder, J., Holtgreve-Grez, H., Jauch, A. & de Lange, T. DNA ligase IV-dependent NHEJ of deprotected mammalian telomeres in G1 and G2. Curr. Biol. 12, 1635–1644 (2002).
Colgin, L.M. & Reddel, R.R. Telomere maintenance mechanisms and cellular immortalization. Curr. Opin. Genet. Dev. 9, 97–103 (1999).
Bryan, T.M., Englezou, A., Gupta, J., Bacchetti, S. & Reddel, R.R. Telomere elongation in immortal human cells without detectable telomerase activity. EMBO J. 14, 4240–4248 (1995).
Bryan, T.M., Englezou, A., Dalla-Pozza, L., Dunham, M.A. & Reddel, R.R. Evidence for an alternative mechanism for maintaining telomere length in human tumors and tumor-derived cell lines. Nat. Med. 3, 1271–1274 (1997).
Perrem, K. et al. Repression of an alternative mechanism for lengthening of telomeres in somatic cell hybrids. Oncogene 18, 3383–3390 (1999).
Dunham, M.A., Neumann, A.A., Fasching, C.L. & Reddel, R.R. Telomere maintenance by recombination in human cells. Nat. Genet. 26, 447–450 (2000).
Jiang, W.Q. et al. Suppression of alternative lengthening of telomeres by Sp100-mediated sequestration of MRE11/RAD50/NBS1 complex. Mol. Cell. Biol. 25, 2708–2721 (2005).
Cesare, A.J. & Reddel, R.R. Telomere uncapping and alternative lengthening of telomeres. Mech. Ageing Dev. 129, 99–108 (2008).
Wang, R.C., Smogorzewska, A. & de Lange, T. Homologous recombination generates T-loop-sized deletions at human telomeres. Cell 119, 355–368 (2004).
Celli, G.B., Denchi, E.L. & de Lange, T. Ku70 stimulates fusion of dysfunctional telomeres yet protects chromosome ends from homologous recombination. Nat. Cell Biol. 8, 885–890 (2006).
Wu, L. et al. Pot1 deficiency initiates DNA damage checkpoint activation and aberrant homologous recombination at telomeres. Cell 126, 49–62 (2006).
Palm, W., Hockemeyer, D., Kibe, T. & de Lange, T. Functional dissection of human and mouse POT1 proteins. Mol. Cell. Biol. 29, 471–482 (2009).
Cesare, A.J. & Griffith, J.D. Telomeric DNA in ALT cells is characterized by free telomeric circles and heterogeneous t-loops. Mol. Cell. Biol. 24, 9948–9957 (2004).
Nabetani, A., Yokoyama, O. & Ishikawa, F. Localization of hRad9, hHus1, hRad1 and hRad17, and caffeine-sensitive DNA replication at ALT (alternative lengthening of telomeres)-associated promyelocytic leukemia body. J. Biol. Chem. 279, 25849–25857 (2004).
Verdun, R.E., Crabbe, L., Haggblom, C. & Karlseder, J. Functional human telomeres are recognized as DNA damage in G2 of the cell cycle. Mol. Cell 20, 551–561 (2005).
Nakamura, A.J. et al. Both telomeric and non-telomeric DNA damage are determinants of mammalian cellular senescence. Epigenetics Chromatin 1, 6 (2008).
Anderson, M.J., Casey, G., Fasching, C.L. & Stanbridge, E.J. Evidence that wild-type TP53, and not genes on either chromosome I or II, controls the tumorigenic phenotype of the human fibrosarcoma HT1080. Genes Chromosom. Cancer 9, 266–281 (1994).
Bunz, F. et al. Requirement for p53 and p21 to sustain G2 arrest after DNA damage. Science 282, 1497–1501 (1998).
Cohen, S.B. & Reddel, R.R. A sensitive direct human telomerase activity assay. Nat. Methods 5, 355–360 (2008).
Henson, J.D., Neumann, A.A., Yeager, T.R. & Reddel, R.R. Alternative lengthening of telomeres in mammalian cells. Oncogene 21, 598–610 (2002).
Murnane, J.P., Sabatier, L., Marder, B.A. & Morgan, W.F. Telomere dynamics in an immortal human cell line. EMBO J. 13, 4953–4962 (1994).
Bailey, S.M., Cornforth, M.N., Kurimasa, A., Chen, D.J. & Goodwin, E.H. Strand-specific postreplicative processing of mammalian telomeres. Science 293, 2462–2465 (2001).
Yeager, T.R. et al. Telomerase-negative immortalized human cells contain a novel type of promyelocytic leukemia (PML) body. Cancer Res. 59, 4175–4179 (1999).
Perrem, K., Colgin, L.M., Neumann, A.A., Yeager, T.R. & Reddel, R.R. Coexistence of alternative lengthening of telomeres and telomerase in hTERT-transfected GM847 cells. Mol. Cell. Biol. 21, 3862–3875 (2001).
Pickett, H.A., Cesare, A.J., Johnstone, R.L., Neumann, A.A. & Reddel, R.R. Control of telomere length by a trimming mechanism that involves generation of t-circles. EMBO J. 28, 799–809 (2009).
Sprung, C.N., Bryan, T.M., Reddel, R.R. & Murnane, J.P. Normal telomere maintenance in immortal ataxia telangiectasia cell lines. Mutat. Res. 379, 177–184 (1997).
Bernardi, R. & Pandolfi, P.P. Structure, dynamics and functions of promyelocytic leukaemia nuclear bodies. Nat. Rev. Mol. Cell Biol. 8, 1006–1016 (2007).
Griffith, J.D. et al. Mammalian telomeres end in a large duplex loop. Cell 97, 503–514 (1999).
Stansel, R.M., de Lange, T. & Griffith, J.D. T-loop assembly in vitro involves binding of TRF2 near the 3′ telomeric overhang. EMBO J. 20, 5532–5540 (2001).
Verdun, R.E. & Karlseder, J. Replication and protection of telomeres. Nature 447, 924–931 (2007).
Bae, N.S. & Baumann, P.A. RAP1/TRF2 complex inhibits nonhomologous end-joining at human telomeric DNA ends. Mol. Cell 26, 323–334 (2007).
Verdun, R.E. & Karlseder, J. The DNA damage machinery and homologous recombination pathway act consecutively to protect human telomeres. Cell 127, 709–720 (2006).
Crabbe, L., Verdun, R.E., Haggblom, C.I. & Karlseder, J. Defective telomere lagging strand synthesis in cells lacking WRN helicase activity. Science 306, 1951–1953 (2004).
Ding, H. et al. Regulation of murine telomere length by Rtel: an essential gene encoding a helicase-like protein. Cell 117, 873–886 (2004).
Konishi, A. & de Lange, T. Cell cycle control of telomere protection and NHEJ revealed by a ts mutation in the DNA-binding domain of TRF2. Genes Dev. 22, 1221–1230 (2008).
Acknowledgements
We thank T. de Lange for supplying the TRF2ΔBΔM construct used in these experiments, and members of the Children's Medical Research Institute for comments on the manuscript. This work was supported by a US National Science Foundation international research fellowship (0602009), a grant from the Cure Cancer Australia Foundation and a Sir Keith Murdoch fellowship from the American Australian Association (to A.J.C.); a Promina postdoctoral fellowship (to H.A.P.); an Australian Postgraduate Award and a Judith Hyam Memorial Trust Fund for Cancer Research scholarship (to Z.K.) and a Cancer Council New South Wales program grant (to R.R.R.).
Author information
Authors and Affiliations
Contributions
A.J.C., Z.K., S.B.C., C.E.N., H.A.P. and A.A.N. conducted the experiments. A.J.C, A.A.N. and R.R.R. designed the project. A.J.C. and R.R.R. analyzed the data and authored the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9, Supplementary Tables 1 and 2 and Supplementary Methods (PDF 8963 kb)
Rights and permissions
About this article
Cite this article
Cesare, A., Kaul, Z., Cohen, S. et al. Spontaneous occurrence of telomeric DNA damage response in the absence of chromosome fusions. Nat Struct Mol Biol 16, 1244–1251 (2009). https://doi.org/10.1038/nsmb.1725
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1725
This article is cited by
-
Telomouse—a mouse model with human-length telomeres generated by a single amino acid change in RTEL1
Nature Communications (2023)
-
Break-induced replication orchestrates resection-dependent template switching
Nature (2023)
-
Telomere-to-mitochondria signalling by ZBP1 mediates replicative crisis
Nature (2023)
-
Homology directed telomere clustering, ultrabright telomere formation and nuclear envelope rupture in cells lacking TRF2B and RAP1
Nature Communications (2023)
-
Signalling inhibition by ponatinib disrupts productive alternative lengthening of telomeres (ALT)
Nature Communications (2023)