Abstract
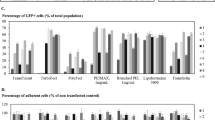
To better characterize lentiviral vector supernatants, we compared three methods of titer assessment. These titer methods include assessment of vector RNA sequences in supernatants, DNA sequences in transduced cells, and vector expression in transduced cells (using a vector which expressed the green fluorescence protein, GFP). For analysis of RNA and DNA, we developed a real-time PCR method for detecting the lentiviral packaging sequence and used this methodology to quantitate the number of vector sequences. Vector expression was assessed by flow cytometric analysis for GFP. As functional titers (DNA and GFP expression titers) are dependent on transduction efficiency, we calculated the titer of a lentiviral vector, RRL-CMV-GFP, after transduction of 293, HeLa, or Mus dunni cells. Genomic DNA was extracted at 4 and 14 days after transduction and the number of vector DNA molecules was determined against a plasmid standard. Of the three cell lines tested, 293 cells provided the highest rate of transduction (PCR estimated DNA titer for RRL-CMV-GFP vector was 2.52 ± 0.25 × 106 molecules/ml at 14 days, and 2.31 ± 0.15 × 106 molecules/ml at 4 days). When titer was calculated based on GFP expression, the highest titer was also obtained on 293 cells (0.26 ± 0.04 × 106 TU/ml at 14 days, and 0.24 ± 0.03 ± 106 TU/ml at 4 days). The titers obtained by GFP expression assay were approximately one log lower than those obtained by DNA analysis suggesting that variability in vector expression may underestimate titer. Measurement of RNA titers directly from vector supernatants against a plasmid standard indicated that the RNA titers are substantially higher than the DNA (~103-fold) and GFP titers (~104-fold). To show that the lentiviral probe and primers could be used for titering a variety of lentiviral vectors, we have also used the real-time PCR method to determine the DNA titers of two other HIV1 derived vectors, RRL-PGK-GFP (6.1 ± 1.4 × 105 molecules/ml), and SMPU-RRE-BN (1.26 ± 0.2 × 106 molecules/ml). We conclude that of the three methods tested, titers assessed by DNA analysis of transduced cells provide the most reliable estimate of functional titers as these are least likely to be influenced by factors, such as defective interfering particles and vector expression levels. The real-time PCR method described offers a reproducible method for lentiviral titering and can be applied to a wide variety of vectors, regardless of transgene.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Vigna E, Naldini L . Lentiviral vectors: excellent tools for experimental gene transfer and promising candidates for gene therapy J Gene Med 2000 2: 308–316
Naldini L, Verma IM . Lentiviral vectors Adv Virus Res 2000 55: 599–609
Buchschacher GL Jr, Wong-Staal F . Development of lentiviral vectors for gene therapy for human diseases Blood 2000 95: 2499–2504
Murdoch B et al. A rapid screening procedure for the identification of high-titer retrovirus packaging clones Gene Therapy 1997 4: 744–749
Sanburn N, Cornetta K . Rapid titer determination using quantitative real-time PCR Gene Therapy 1999 6: 1340–1345
Tafuro S, Zentilin L, Falaschi A, Giacca M . Rapid retrovirus titration using competitive polymerase chain reaction Gene Therapy 1996 3: 679–684
Cheng L, Fu J, Tsukamoto A, Hawley RG . Use of green fluorescent protein variants to monitor gene transfer and expression in mammalian cells Nat Biotechnol 1996 14: 606–609
Riviere I, Brose K, Mulligan RC . Effects of retroviral vector design on expression of human adenosine deaminase in murine bone marrow transplant recipients engrafted with genetically modified cells Proc Natl Acad Sci USA 1995 92: 6733–6737
Onodera M et al. A simple and reliable method for screening retroviral producer clones without selectable markers Hum Gene Ther 1997 8: 1189–1194
Davis JL et al. Retroviral particles produced from a stable human-derived packaging cell line transduce target cells with very high efficiencies Hum Gene Ther 1997 8: 1459–1467
Cashion LM et al. Use of enhanced green fluorescent protein to optimize and quantitate infection of target cells with recombinant retroviruses Biotechniques 1999 26: 924–926
Naldini L et al. Efficient transfer, integration, and sustained long-term expression of the transgene in adult rat brains injected with a lentiviral vector Proc Natl Acad Sci USA 1996 93: 11382–11388
Miller AD . Retrovirus packaging cells Hum Gene Ther 1990 1: 5–14
Yamada K et al. Functional correction of fanconi anemia group C hematopoietic cells by the use of a novel lentiviral vector Mol Ther 2001 3: 485–490
Naldini L et al. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector Science 1996 272: 263–267
Dull T et al. A third-generation lentivirus vector with a conditional packaging system J Virol 1998 72: 8463–8471
Kirkwood TB, Bangham CR . Cycles, chaos, and evolution in virus cultures: a model of defective interfering particles Proc Natl Acad Sci USA 1994 91: 8685–8689
Le Doux JM, Morgan JR, Snow RG, Yarmush ML . Proteoglycans secreted by packaging cell lines inhibit retrovirus infection J Virol 1996 70: 6468–6473
White SM et al. Lentivirus vectors using human and simian immunodeficiency virus elements J Virol 1999 73: 2832–2840
Chen J et al. Packaging cell line DNA contamination of vector supernatants: implication for laboratory and clinical research Virology 2001 282: 186–197
Acknowledgements
We thank Dr Donald Kohn for the SMPU-RRE-BN vector construct and Dr Rafat Abonour and Dr Christie Traycoff for helpful comments on the manuscript. L Sastry is supported by the National Institutes of Health, National Research Service Award number 1T32 HL07910, Basic Science Studies on Gene Therapy of Blood Disease. The Indiana University Vector Production Facility is a NIH-designated National Gene Vector Laboratory (U42 RR11148) and this work was supported in part by a Core Centers for Excellence in Molecular Hematology (CCEMH) grant (PHS P50 DK 49218) and a core laboratory supporting PHS P01 HL53586.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sastry, L., Johnson, T., Hobson, M. et al. Titering lentiviral vectors: comparison of DNA, RNA and marker expression methods. Gene Ther 9, 1155–1162 (2002). https://doi.org/10.1038/sj.gt.3301731
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301731
Keywords
This article is cited by
-
ANGPTL4 is a potential driver of HCV-induced peripheral insulin resistance
Scientific Reports (2023)
-
Quantitation and integrity evaluation of RNA genome in lentiviral vectors by direct reverse transcription-droplet digital PCR (direct RT-ddPCR)
Scientific Reports (2023)
-
Generation of CRISPR–Cas9-mediated genetic knockout human intestinal tissue–derived enteroid lines by lentivirus transduction and single-cell cloning
Nature Protocols (2022)
-
A guide in lentiviral vector production for hard-to-transfect cells, using cardiac-derived c-kit expressing cells as a model system
Scientific Reports (2021)
-
Lentivirus expressing shRNAs inhibit the replication of contagious ecthyma virus by targeting DNA polymerase gene
BMC Biotechnology (2020)