Summary
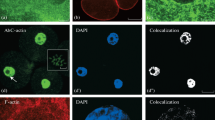
The amniotic epithelium constitutes a diffusion barrier controlling the passage of solutes and water between the aminotic cavity and maternal circulation. With the present immunocytochemical approach, we have shown that several major components of the cytoskeleton, i.e., actin, α-actinin, spectrin and ezrin, are preferentially associated with the apical and lateral cell surfaces of the human amniotic epithelium. Keratins are distributed throughout the entire cytoplasm, whereas vimentin mainly forms a perinuclear scaffold. These findings indicate a role of the various components of the cytoskeleton in the structural integrity and modulation of cell shape and junctional permeability.
Similar content being viewed by others
References
Bartels H, Wang T (1983) Intercellular junctions in the human fetal membranes. Anat Embryol 166:103–120
Bennett V (1985) The membrane skeleton of human erythrocytes and its implications for more complex cells. Ann Rev Biochem 54:273–304
Bretscher A (1983) Purification of an 80000-dalton protein that is a component of the isolated microvillus cytoskeleton and its localisation in non-muscles cells. J Cell Biol 97:425–432
Bretscher A (1989) Rapid phosphorylation and reorganization of ezrin and spectrin accompany morphological changes induced in A-431 cells by epidermal growth factor. J Cell Biol 108:921–930
Burgess DR (1982) Reactivation of intestinal epithelial cell brush border motility: ATP-dependent contraction via a terminal web contractile ring. J Cell Biol 95:853–863
Drenckhahn D (1988) Zytoskelett und Zelldifferenzierung. Verh Dtsch Ges Path 72:10–29
Drenckhahn D, Dermietzel R (1988) Organization of the actin filament cytoskeleton in the intestinal brush border: a quantitative and qualitative immunoelectron microscope study. J Cell Biol 107:1037–1048
Drenckhahn D, Franz H (1986) Identification of actin, α-actinin, and vinculin containing plaques at the lateral membrane of epithelial cells. J Cell Biol 102:1843–1852
Drenckhahn D, Gröschel-Stewart U (1980) Localization of myosin, actin, and trypomyosin in rat intestinal epithelium. Immunocytochemical studies at the light- and electron microscopic levels. J Cell Biol 86:475–482
Drenckhahn D, Wagner J (1986) Stress fibers in the splenic sinus endothelium in situ: molecular structure, relationships to the extracellular matrix and contractility. J Cell Biol 102:1738–1747
Drenckhahn D, Hofmann HD, Mannherz HG (1983) Evidence for the association of villin with core filaments and rootlets of intestinal epithelial microvilli. Cell Tissue Res 228:409–414
Drenckhahn D, Schäfer T, Prinz M (1985) Actin, myosin and associated proteins in the vertebrate auditory and vestibular organ: immunocytochemical and biochemical studies. In: Drescher DG (ed) Auditory biochemistry. Thomas, Springfield, Ill, USA
Franke H, Estel C (1978) Untersuchungen über die Ultrastruktur und Permeabilität des Amnions unter besonderer Berücksichtigung mikrofilamentärer und mikrotubulärer Strukturen. Arch Gynäk 225:319–338
Gould KL, Cooper JA, Bretscher A, Hunter Z (1986) The proteintyrosin kinase substrate, p 81, is homologous to a chicken microvillar core protein. J Cell Biol 102:660–669
Gröschel-Stewart U, Drenckhahn D (1982) Muscular and cytoplasmic contractile proteins. Biochemistry, immunology, structural organization. J Collagen Rel Res 2:381–463
King BF (1982) Cell surface specializations and intercellular junctions in human amniotic epithelium: an electron microscopic and freeze-fracture study. Anat Rec 203:73–82
Koob R, Kraemer D, Trippe G, Aebi U, Drenckhahn D (1990) Association of kidney and parotid Na+,K+-ATPase microsomes with actin and analogs of spectrin and ankyrin. Eur J Cell Biol 53:641–651
Krantz KE, Panos TC, Evans J (1962) Physiology of maternal-fetal relationship through the extracorporeal circulation of the human placenta. Am J Obstet Gynecol 83:1214–1227
Levine J, Willard M (1981) Fodrin: axonally transported polypeptides associated with the internal periphery of many cells. J Cell Biol 90:631–643
Madara JL (1989) Loosening tight junctions. Lessons from the intestine. J Clin Invest 83:1089–1094
Mayor HD, Hampton JC, Rosario B (1961) A simple method for removing the resin from epoxy-embedded tissue. J Biophys Biochem Cytol 9:909–910
Owaribe K, Kodama R, Eguchi G (1981) Demonstration of contractibility of circumferential actin bundles and its morphogenic significance in pigmented epithelium in vitro and in vivo. J Cell Biol 90:507–514
Petry G (1980) “Autodesmosomen”, desmosomale Kontakte von Teilen derselben Zelle im menschlichen Chorion laeve und Amnion. Eur J Cell Biol 23:129–136
Regauer S, Franke WW, Virtanen I (1985) Intermediate filament cytoskeleton of amnion epithelium and cultured amnion epithelial cells: expression of epidermal cytokeratins in cells of a simple epithelium. J Cell Biol 100:997–1009
Schmidt W, Pfaller K, Schwarzfurtner H (1982) Licht- und elektronenmikroskopische Untersuchungen an den Eihäuten des Menschen. I. Amnion und Zwischenschicht. Zbl Gynäkol 104:385–396
Schnittler HJ, Wilke A, Gress T, Suttorp N, Drenckhahn D (1990) Role of actin and myosin in the control of paracellular permeability in pig, rat and human vascular endothelium. J Physiol 431:379–401
Seeds AE (1980) Current concepts of amniotic fluid dynamics. Am J Obstet Gynecol 138:575–586
Starger JH, Brown WE, Goldmann AE, Goldmann RD (1978) Biochemical and immunological analysis of rapidly purified 10 nm filaments from baby hamster kidney (BHK-21) cells. J Cell Biol 78:93–109
Thomas CE (1965) The ultrastructure of human amnion epithelium. J Ultrastruct Res 13:65–84
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wolf, H.J., Schmidt, W. & Drenckhahn, D. Immunocytochemical analysis of the cytoskeleton of the human amniotic epithelium. Cell Tissue Res 266, 385–389 (1991). https://doi.org/10.1007/BF00318194
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00318194