Summary
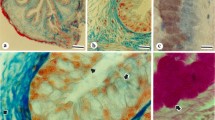
The location of endogenous avidin was studied cytochemically in the magnum tissue of the oviduct of laying hens. Two methods, based on an interaction of avidin-biotin with biotin hydrazide-peroxidase (B-HRP) as an affinity reagent, and on an immunoperoxidase technique, were tested by morphological analysis. The data obtained by both methods showed that in the magnum B-HRP is a strictly substitutive reagent for endogenous avidin.
Avidin was clearly demonstrated in large amounts in the secretory granules of some epithelial cells and tubular gland cells, but was absent from mucous cells, the goblet cells, which had been believed to be the location of avidin production, and from ciliated cells. These granules had previously been demonstrated by both electron-microscopic cytochemical techniques. Especially in acinar cells, they were nonhomogeneous with a speckled core and a dense peripheral part. They ranged in size from 500 to 2200nm in diameter in the gland and 180 to 720 nm in the epithelium. Columnar epithelial cells containing avidin granules had a strong resemblance to those of the protodifferentiated tubular gland cells in the magnum of chicks pretreated with daily estrogen or estrogen plus progesterone, and might have migrated towards the acinus as substitutional secretory cells.
Therefore, the acinar cells of the magnum, considered to be composed of several secretory protein-producing systems, are dependent on estrogen and/or progesterone in the oviduct of the laying hen.
Similar content being viewed by others
References
Cox RF, Sauerwein H (1970) Studies on the mode of action of progesterone on chicken oviduct epithelium. Exp Cell Res 61:79–90
Fertuck HC, Newstead JD (1970) Fine structural observations on magnum mucosa in quail and hen oviducts. Z Zellforsch 103:447–459
Heitzmann H, Richards FM (1974) Use of the avidin-biotin complex for specific staining of biological membranes in electron microscopy. Proc Nat Acad Sci USA 71:3537–3541
Kami K, Yasuda K (1982) Biotinylated-enzymes affinity cytochemistry to demonstrate endogenous avidin in hen oviducts: Preliminary study. Okajimas Fol Anat Jpn 58:1289–1304
Kami K, Yasuda K (1983a) Tubular gland cell cytodifferentiation and cytochemical demonstration of avidin of chick oviduct. Acta Anat Nippon 58:396
Kami K, Yasuda K (1983b) Cytochemical demonstration of endogenous avidin found in secretory granules of the laying hen oviduct using immunoperoxidase methods: Preliminary report. Histochem J (under contribution)
Kami K, Suzuki T, Mitsui T (1976) Immunohistochemical observations of hen egg white trypsin inhibitor. Acta Histochem Cytochem 9:12
Kami K, Mitsui T, Suzuki T (1978) Distribution of lysozyme in tissues and cells of the domestic fowl. Acta Histochem Cytochem 11:107
Kami K, Suzuki T, Igarashi J, Mitsui T (1979) Immunohistochemical demonstration of lysozyme in tissues and blood cells of domestic fowl. Okajimas Fol Anat Jpn 55:313–328
Kellokumpu-Lehtinen P, Jokelainen PT, Tuohimaa P (1976) Early cytodifferentiation of the chick oviduct epithelium by progesterone. J Ultrastruct Res 54:190–201
Kohler PO, Grimley PM, O'Malley BW (1968) Protein synthesis: Differential stimulation of cell-specific proteins in epithelial cells of chick oviduct. Science 160:86–87
Makita T, Kiwaki S, Sandborn EB (1973) Secretion granules in the ciliated cells of the avian oviduct. Arch Histol Jpn 35:209–224
Nordback I, Ratia J, Tuohimaa P (1980) Avidin synthesis in vitro by chick oviduct after a short induction with progesterone or actinomycin D in vitro. J Steroid Biochem 12:283–286
O'Malley BW (1967) In vitro hormonal induction of a specific protein (avidin) in chick oviduct. Biochemistry 6:2546–2551
O'Malley BW, McGuire WL (1968) Studies on the mechanism of action of progesterone in regulation of the synthesis of specific protein. J Clin Invest 47:654–664
Palmiter RD, Wrenn JT (1971) Interaction of estrogen and progesterone in chick oviduct development: III. Tubular gland cell cytodifferentiation. J Cell Biol 50:598–615
Palmiter RD, Christensen AK, Schimke RT (1970) Organization of polysomes from pre-existing ribosomes in chick oviduct by a secondary administration of either estradiol or progesterone. J Biol Chem 245:833–845
Sanderson CJ, Wilson DV (1971) A simple method for coupling proteins to insoluble polysaccharides. Immunology 20:1061–1065
Schrader MR, Birnbaumer ME, Hughes MR, Weigl NL, Grody WE, O'Malley BW (1981) Studies in the structure and function of the chick progesterone receptor. Rec Prog Horm Res 37:583–633
Suzuki T, Nagato Y (1980) Immunohistochemical investigation of chick oviduct using a GMA-Quetol 523 embedding method. Acta Histochem Cytochem 13:486–498
Suzuki T, Takano K, Yasuda K (1970) Immunohistological investigation on the serous gland of chick oviduct: I. On the localization of ovalbumin. Acta Histochem Cytochem 3:114–121
Tuohimaa P (1975) Immunofluorescence demonstration of avidin in the immature chick oviduct epithelium after progesterone. Histochemistry 44:95–101
Wigham T, Nordback I, Ratia J, Tuohimaa P (1980) Progesterone-dependent and -independent avidin production in chick oviduct organ culture. Med Biol 58:14–17
Yamashita S, Yamamoto N, Yasuda K (1976) Purification of peroxidase labeled antibody by using DEAE-cellulose chromatography. Acta Histochem Cytochem 9:227–233
Yasuda K, Kami K (1981) Application of avidin-biotin interaction to the histochemical and immunohistochemical studies. Acta Anat Nippon 56:261–262
Yasuda K, Yamamoto N, Yamashita S (1981) Use of peroxidase-avidin conjugate for the demonstration of intracellular antigen. Experientia 37:306–307
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kami, K., Yasuda, K. Endogenous avidin found in the magnum gland of the hen oviduct. Cell Tissue Res. 234, 439–450 (1983). https://doi.org/10.1007/BF00213780
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00213780