Abstract
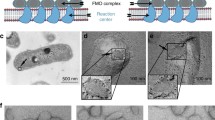
The photosynthetic membranes of the five bchl b-containing bacteria Ectothiorhodospira halochloris, E. abdelmalekii, Rhodopseudomonas viridis, R. sulfoviridis and Thiocapsa pfennigii have been investigated by electron microscopy and digital image analysis. All five species have the photosynthetic complexes hexagonally arrayed in the membrane with lattice spacings close to 13 nm, except for R. sulfoviridis and T. pfennigii which display somewhat smaller (∼12.5 nm) lattice spacings.
Correlation averaging which imposes less stringent requirements on the lattice perfection than conventional Fourier filtration techniques has been employed to elucidate the structure of the photosynthetic complexes. Their basic organization, i.e. a ring, probably containing the light-harvesting (LH) polypeptides, surrounding a core (the “reaction centre”) appears to be almost identical for all species under scrutiny. Despite a resolution of ∼1.6 nm, however, little further significant substructure can be deduced from the averages; possible reasons for the “blurred” appearance of the LH-ring and absence of any subdivision in the reaction centre are discussed along with strategies aimed at obtaining a more detailed model of the molecular architecture of the photosynthetic membranes.
Similar content being viewed by others
References
Brunisholz RA, Jay F, Suter F, Zuber H (1982) The complete primary structure of the light harvesting polypeptides from various photosynthetic bacteria. In: Abstracts FEMS symposium on function and biosynthesis of energy-conserving membranes. Freiburg
Drews G, Giesbrecht P (1965) Die Thylakoidstrukturen von Rhodopseudomonas spec. Arch Mikrobiol 52:242–250
Drews G, Giesbrecht P (1966) Rhodopseudomonas viridis, nov. spec., ein neu isolierters obligat phototrophes Bakterium. Arch Mikrobiol 53:255–262
Drews G, Oelze J (1981) Organization and differentiation of membranes of phototrophic bacteria. Adv Microb Physiol 22:1–92
Eimhjellen KE, Steensland H, Traetteberg GJ (1967) A Thiocaccus sp. nov. gen., its pigments and internal membrane system. Arch Mikrobiol 59:82–92
Engelhardt H, Klemme JH (1978) Characterization of an allosteric, nucleotide-unspezific glutamate dehydrogenase from Rhodopseudomonas sphaeroides. FEMS Microbiol Lett 3:287–290
Frank J, Goldfarb W, Eisenberg D, Baker TS (1978) Reconstruction of glutamine synthetase using computer averaging. Ultramicroscopy 3:283–290
Giesbrecht P, Drews G (1966) Über die Organisation und die makromolekulare Architektur der Thylakoide “lebender” Bakterien. Arch Mikrobiol 54:297–330
Gross H (1980) Ultrahigh vacuum freeze-fracturing at-196°C and decoration of specific sites of paracrystalline membranes. In: Baumeister W, Vogell W (eds) Electron microscopy at molecular dimensions. Springer, Berlin Heidelberg New York, pp 71–79
Imhoff JF, Trüper HG (1977) Ectothiorhodospira halochloris sp. nov., a new extremely halophilic phototrophic bacterium containing bacteriochlorophyll b. Arch Microbiol 114: 115–121
Imhoff JF, Trüper HG (1981) Ectothiorhodospira abdelmalekii sp. nov., a new halophilic and alkaliphilic phototrophic bacterium. Zbl Bakt Hyg I. Abt Orig C2:228–234
Keppen OI, Gorlenko VM (1975) A new species of purple budding bacteria containing bacteriochlorophyll b. Mikrobiologiya 44:258–263
Kühlbrandt W, Wehrli E, Thaler T, Mühlethaler K (1982) Structures of two regular arrays of photosynthetic complexes. Proc 10th Intl Congr Electron Microscopy Vol 3:45–46
Meyer R, Snozzi M, Bachofen R (1981) Freeze fracture studies of reaction centers from Rhodospirillum rubrum in chromatophores and liposomes. Arch Microbiol 130:125–128
Miller KR (1979) Structure of a bacterial photosynthetic membrane. Proc Natl Acad Sci USA 76:6415–6419
Miller KR (1982) Three-dimensional structure of a photosynthetic membrane. Nature 300:53–55
Monger TG, Parson WW (1977) Singlett-triplett fusion in Rhodopseudomonas sphaeroides chromatophores. A probe of the organization of the photosynthetic apparatus. Biochim Biophys Acta 460:393–407
Neutzling O, Trüper HG (1982) Assimilatory sulfur metabolism in Rhodopseudomonas sulfoviridis. Arch Microbiol 133:145–148
Oelze J, Golecki JR (1975) Properties of reaction center depleated membranes of Rhodspirillum rubrum. Arch Microbiol 102:59–64
Oyewole SH, Holt SC (1976) Structure and composition of intracytoplasmic membranes of Ectothiorhodospira mobilis. Arch Microbiol 107:167–182
Saxton WO, Pitt TJ, Horner M (1979) Digital image processing: the Semper system. Ultramicroscopy 4:343–354
Saxton WO, Baumeister W (1982) The correlation averaging of a regularly arranged bacterial cell envelope protein. J Microscopy 127:127–138
Saxton WO, Koch TL (1982) Interactive image processing with an off-line minicomputer: organization, performance and applications. J Microscopy 127:69–83
Takemoto JY, Peters J, Drews G (1982) Crosslinking of photosynthetic membrane polypeptides of Rhodopseudomonas capsulata. FEBS Lett 142:227–230
Tiede DM, Dutton PL (1981) Orientation of the primary quinone of bacterial photosynthetic reaction centers contained in chromatophore and reconstituted membranes. Biochim Biophys Acta 637:278–290
Ueki T, Kataoka M, Mitsui T (1976) Structural order in chromatophore membranes of Rhodospirillum rubrum. Nature 262:809–810
Wehrli E, Kübler O (1980) The two-dimensional lattice of the photosynthetic membrane of Rhodopseudomonas viridis. In: Baumeister W, Vogell W (eds) Electron microscopy at molecular dimensions. Springer, Berlin Heidelberg New York, pp 48–56
Wrigley NG (1968) The lattice spacing of crystalline catalase as an internal standard of length in electron microscopy. J Ultrastruct Res 24:454–464
Zuber H (1982) Structural studies on the light-harvesting pigmentprotein complexes from cyanobacteria and photosynthetic (purple) bacteria. Abstract IV. Intl Symp Photosynth Prok, Bombannes, France, p C36
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Engelhardt, H., Baumeister, W. & Saxton, W.O. Electron microscopy of photosynthetic membranes containing bacteriochlorophyll b . Arch Microbiol 135, 169–175 (1983). https://doi.org/10.1007/BF00414474
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00414474