Abstract
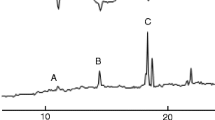
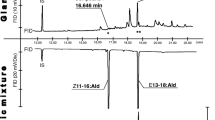
The sex pheromone ofEarias insulana, (E,E)-10,12-hexadecadienal, may trimerize extensively to form a crystalline trioxane derivative. The structure of the trimer was deduced from its CI-MS and NMR spectra. Capillary GC analysis resulted in the thermal decomposition of the trimer to the monomer. This process could be studied on a 2-m packed column under specific conditions. A convenient separation between the pheromone and its trimer was achieved by TLC. The trimer was inactive in the field, and it has a harmful effect on the performance of the polyethylene dispenser. Material which contains large amounts of the trimer is unsuitable for field use, even if applied at high dosage. The pheromone should be analyzed by NMR or TLC in addition to GC in order to detect the presence of its trimer. The trimerization process is catalyzed by acid which should therefore be completely eliminated from the storing vessels.
Similar content being viewed by others
References
Buchanan, J.G., Hughes, N.A., McQuimN, F.J., andSwan, G.A. 1965. Aliphatic compounds, aldehydes and ketones, p. 21,in Rodd's Chemistry of Carbon Compounds, 2nd ed, Vol. Ic. Elsevier, Amsterdam.
Francke, A., andWozelka, H. 1912. Über die Polymerisation einiger Aldehyde der Reihe CnH2nO.Monatshefte 33:349–362.
Hall, D.R., Beevor, P.S., Lester, R., andNesbitt, B.F. 1980. (E,E)-10,12-hexadecadienal: A component of the female sex pheromone of the spiny bollworm,Earias insulana (Boisd.) (Lepidoptera, Noctuidae).Experientia 36:152–153.
Jungnickel, J.L., andReilly, C.A. 1965. Nuclear magnetic resonance studies on geometrical isomers of paraldehyde and other substituted trioxanes.J. Mol. Spectrosc. 16:135–157.
Kehat, M., Gothilf, S., Dunkelblum, E., andGreenberg, S. 1981a. Captures ofEarias insulana males in water traps and dry funnel traps baited with synthetic pheromone or virgin females.Phytoparasitica 9:149–151.
Kehat, M., Gothilf, S., Dunkelblum, E., andMazor, M. 1981b. Sex pheromone traps as a potential means of improving control programs for the spiny bollworm,Earias insulana.Phytoparasitica 9:191–196.
Klug, J.T.,Skorka, J., andShani, A. 1982. A short synthesis of (E,E)-10,12-hexadecadienal: The main component of the female sex pheromone of the spiny bollworm (Earias insulana).Chem. Ind. pp. 372–373.
Author information
Authors and Affiliations
Additional information
Boisduval, Lepidoptera: Noctuidae.
Contribution from the Agricultural Research Organization (ARO), No. 675-E, 1983 series.
Rights and permissions
About this article
Cite this article
Dunkelblum, E., Kehat, M., Klug, J.T. et al. Trimerization ofEarias insulana sex pheromone, (E,E)-10,12-Hexadecadienal, A phenomenon affecting trapping efficiency. J Chem Ecol 10, 421–428 (1984). https://doi.org/10.1007/BF00988089
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00988089